Blog by Deanna Vaughn, PT, DPT who practices at Core and Pelvic Physical Therapy Clinic in Conway, Arkansas, this article was originally located at https://whatsupdownthere.info/colorectal-cancer-the-gut-and-the-butt/.
Colorectal cancer refers to cancerous cells within the colon or rectum. Need a quick anatomy review? Keep reading then!
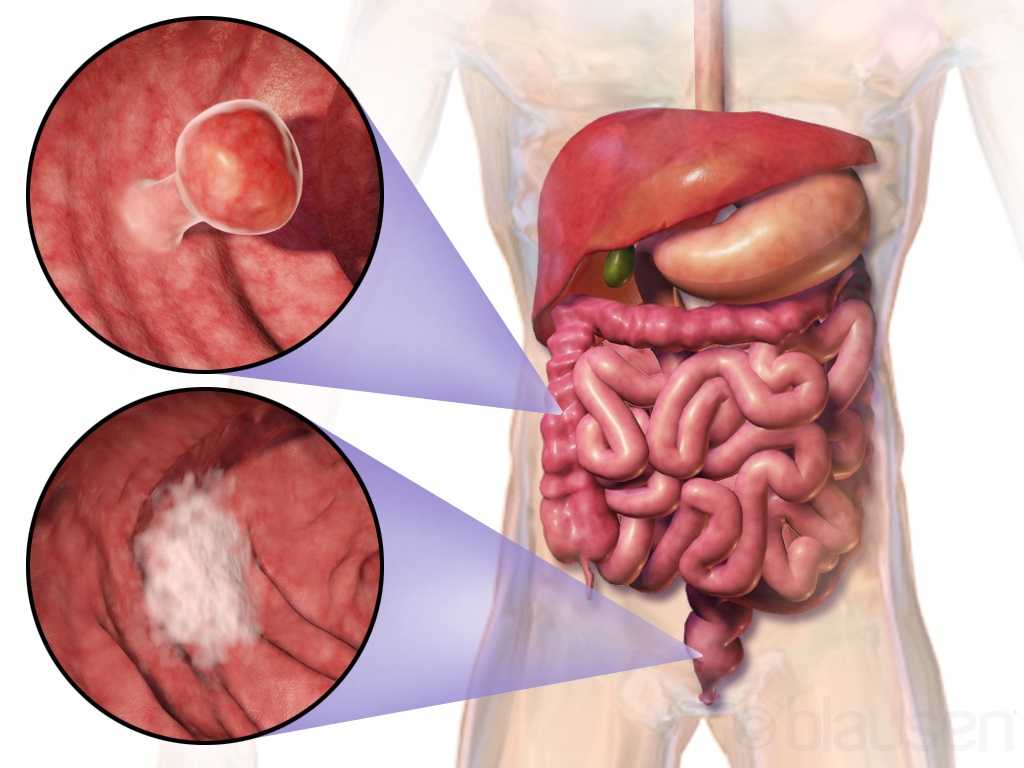
The colon is another name for the large intestine, which is the long tube (nearly 5 FEET!) surrounding the small intestines (that snaky, jumbled tube in the middle of our bodies, which you can see below in the picture). It’s comprised of segments: the cecum (the little pouch that joins the small intestine to the large intestine) in the right lower abdomen, the ascending colon starting at the right lower part of your abdomen (coming off the cecum), and up to about the right side of your ribcage; the transverse colon that loops underneath the stomach and ribcage from right to left; the descending colon that extends down from the left side of your ribcage to the lower part of your left abdomen; and then the sigmoid colon that loops (in an s-shape) along the lower abdomen to the center of the body. At the end of the colon is the rectum, which pretty much connects the colon to the actual anus/anal opening for wastes to leave the body.
That being said, colorectal cancer can affect any part or segment of the colon and the rectum. If you have a family history of colorectal cancer, or if you have an inflammatory bowel disease (like Crohn’s disease or ulcerative colitis), then you may be at a higher risk for colorectal cancer. Other risk factors are the same for virtually any other health condition – genetics, no regular physical activity, poor diet, tobacco use, high alcohol consumption, etc.
So how would we know if it’s colorectal cancer – or precancerous cells, and how do we decrease our risk?
That’s where screening comes into play! Just like how someone may see their gynecologist annually and undergo the PAP smear every 1-3 years to check for any gynecological cancer (like cervical or labial cancer), someone may see their colorectal or gastrointestinal (GI) provider to check for colorectal cancer or disorders. Regular screening takes place around age 45 (although a person may be screened earlier if they are at higher risk or had a previous history of cancer).
What does screening look like?
There are a few tests that screen for colorectal cancer. These tests include stool tests, flexible sigmoidoscopy, and colonoscopy.
Stool tests – This pretty much involves you taking a sample of your stool via test kit provided to you, and returning it to your doctor/lab, where your stool is checked for any blood or other abnormal findings.
Flexible sigmoidoscopy – A thin, short tube with a light is inserted into the rectum. This allows your doctor to see any polyps or cancer within the rectum and lower part of the colon.
Colonoscopy – This is like the sigmoidoscopy, but with a longer tube. The longer tube allows your doctor to check for polyps/cancer inside the rectum and the entire length of the colon. Your doctor can also remove some polyps during this procedure if indicated.
Most people without any symptoms, abnormal findings or outstanding personal or family history of colorectal cancer will have these screening tests performed anywhere from 5-10 years.
What are the symptoms?
This is not an exhaustive list, but some symptoms may include:
- Bleeding, pain, and/or discomfort within the rectum/anus
- Blood in stool
- Abdominal pain and bloating
- Nausea/vomiting
- Difficulty or incomplete bowel evacuation
- Hemorrhoids
- Altered bowel habits (such as sudden constipation, diarrhea, change in stool consistency)
Now what are our treatment options?
Besides preventative measures – such as getting regular physical activity, improving our diet, etc., treatment looks similar to any other cancer treatment. This may look like chemotherapy, radiation therapy, immunotherapy, and/or surgery. Surgery may be indicated to remove polyps/tumors, or parts of the colon or rectum to eliminate cancerous growths. Thankfully though, regular screening of the colorectal region can find precancerous/cancerous cells early. Oftentimes, such as during a colonoscopy, your colorectal provider may go ahead and remove polyps that are abnormal or deemed precancerous at that time!
Now what about pelvic physical therapy? Can it possibly help?
Well, this is another condition (like Pelvic Congestion Syndrome in the previous blog post), where pelvic physical therapy is not the initial go-to or main treatment option. Individuals with colorectal cancer vary in several ways depending on staging/severity and overall health. Once again, pelvic therapy is a nice resource to utilize if you’re needing or wanting ways to manage your bowel symptoms.
Ways that pelvic PT CAN help may include: Teaching appropriate toileting – positioning to straighten out the anorectal angle and allow stool to pass more easily from the rectum; mechanics, such as exhaling smoothly when pushing for a bowel movement to prevent straining; Improving pelvic floor muscle function (strength, endurance, coordination) so that your body can delay defecation as needed and calm down bowel urges; and overall promoting health bowel habits by supporting your nutrition and keeping bowel movements regular.
Whether or not you (or someone you know) have colorectal cancer, developing healthy and safe bowel habits is key to a better quality of life. Working with your doctor and/or your team of providers is important in making sure your needs are addressed, but feel free to reach out to your local pelvic PT if you want more resources or guidance – even things like, “So, how SHOULD I be pooping??”
References & Resources
Brenner H, Chen C. The colorectal cancer epidemic: challenges and opportunities for primary, secondary and tertiary prevention. Br J Cancer. 2018;119(7):785-792. doi:10.1038/s41416-018-0264-x
https://www.cancer.org/cancer/colon-rectal-cancer.html
https://my.clevelandclinic.org/health/diseases/14501-colorectal-colon-cancer
Kuipers EJ, Grady WM, Lieberman D, et al. Colorectal cancer. Nat Rev Dis Primers. 2015;1:15065. Published 2015 Nov 5. doi:10.1038/nrdp.2015.65
Leslie A, Steele RJC. Management of colorectal cancerPostgraduate Medical Journal 2002;78:473-478. http://dx.doi.org/10.1136/pmj.78.922.473
Mármol I, Sánchez-de-Diego C, Pradilla Dieste A, Cerrada E, Rodriguez Yoldi MJ. Colorectal Carcinoma: A General Overview and Future Perspectives in Colorectal Cancer. Int J Mol Sci. 2017;18(1):197. Published 2017 Jan 19. doi:10.3390/ijms18010197
You YN, Lee LD, Deschner BW, Shibata D. Colorectal Cancer in the Adolescent and Young Adult Population. JCO Oncol Pract. 2020;16(1):19-27. doi:10.1200/JOP.19.00153
Curing cancer but not addressing life-altering complications can be compared to feeding the homeless on Thanksgiving but turning your back on them the rest of the year. We love hearing positive outcomes of a surgery, but we are not always aware of what happens beyond that. Colorectal cancer is often treated by colectomy, and sometimes the survivor of cancer is left with urological or sexual dysfunction, small bowel obstruction, or pelvic lymphedema.
 Panteleimonitis et al., (2017) recognized the prevalence of urological and sexual dysfunction after rectal cancer surgery and compared robotic versus laparoscopic approaches to see how each impacted urogenital function. In this study, 49 males and 29 females underwent laparoscopic surgery, and 35 males and 13 females underwent robotic surgery. Prior to surgery, 36 men and 9 women were sexually active in the first group and 13 men and 4 women were sexually active in the latter group. Focusing on the male results, male urological function (MUF) scores were worse pre-operatively in the robotic group for frequency, nocturia, and urgency compared to the laparoscopic group. Post-operatively, urological function scores improved in all areas except initiation/straining for the robotic group; however, the MUF median scores declined in the laparoscopic group. Regarding male sexual function (MSF) scores for libido, erection, stiffness for penetration and orgasm/ ejaculation, the mean scores worsened in all areas for the laparoscopic group but showed positive outcomes for the robotic group. In spite of limitations of the study, the authors concluded robotic rectal cancer surgery may afford males and females more promising urological and sexual outcomes as robotic.
Panteleimonitis et al., (2017) recognized the prevalence of urological and sexual dysfunction after rectal cancer surgery and compared robotic versus laparoscopic approaches to see how each impacted urogenital function. In this study, 49 males and 29 females underwent laparoscopic surgery, and 35 males and 13 females underwent robotic surgery. Prior to surgery, 36 men and 9 women were sexually active in the first group and 13 men and 4 women were sexually active in the latter group. Focusing on the male results, male urological function (MUF) scores were worse pre-operatively in the robotic group for frequency, nocturia, and urgency compared to the laparoscopic group. Post-operatively, urological function scores improved in all areas except initiation/straining for the robotic group; however, the MUF median scores declined in the laparoscopic group. Regarding male sexual function (MSF) scores for libido, erection, stiffness for penetration and orgasm/ ejaculation, the mean scores worsened in all areas for the laparoscopic group but showed positive outcomes for the robotic group. In spite of limitations of the study, the authors concluded robotic rectal cancer surgery may afford males and females more promising urological and sexual outcomes as robotic.
Husarić et al., (2016) considered the risk factors for adhesive small bowel obstruction (SBO) after colorectal cancer colectomy, as SBO is a common morbidity that causes a decrease in quality of life. They performed a retrospective study of 248 patients who underwent colon cancer surgery, and 13.7% of all the patients had SBO. Thirty (14%) of the 213 males and 9 (12.7%) of the 71 females had SBO; consequently, they found patients being >60 years old was a more significant risk factor than sex regarding occurrence of SBO. The authors concluded a Tumor-Node Metastasis stage of >3 and immediate postoperative complications were found to be the greatest risk factors for SBO.
Vannelli et al., (2013) explored the prevalence of pelvic lymphedema after lymphadenectomy in patients treated surgically for rectal cancer. Five males and 8 females were examined one week before and 12 months after being discharged from the hospital. All 9 of the patients (4 males, 5 females) with extra-peritoneal cancer exhibited lymphedema via MRI, but the 4 (1 male, 3 females) patients with intra-peritoneal cancer had none. The authors concluded pelvic lymphedema can be elusive after rectal surgery, but pelvic disorders persist and patients should be routinely examined for it.
Obviously saving a life is the primary goal when it comes to cancer. But just like caring for the destitute for one day doesn’t cure a lifetime of hunger, ignoring the negative post-surgical sequelae of a colectomy prevents a cancer survivor from living a healthy life. Herman & Wallace offers two pelvic floor oncology courses, “Oncology and the Male Pelvic Floor” and "Oncology and the Female Pelvic Floor" , which address how pelvic cancers affect the quality of life of our patients and how practitioners can make a positive impact.
Panteleimonitis, S., Ahmed, J., Ramachandra, M., Farooq, M., Harper, M., & Parvaiz, A. (2017). Urogenital function in robotic vs laparoscopic rectal cancer surgery: a comparative study. International Journal of Colorectal Disease, 32(2), 241–248. http://doi.org/10.1007/s00384-016-2682-7
Husarić E., Hasukić Š, Hotić N, Halilbašić A, Husarić S, Hasukić I. (2016). Risk factors for post-colectomy adhesive small bowel obstruction. Acta
Ulcerative Colitis (UC) dramatically effects a patient’s livelihood. UC is often confused with Crohn’s Disease, another major inflammatory bowel disease. While they do differ in origin, both diseases share similar symptoms, such as blood in a patient’s stool. Furthermore, like Crohn’s Disease, UC tends to affect young people (those between the ages of fifteen and thirty).

Chronic and often severe, UC has no known cure and, in rare cases, can even be life-threatening to the patient.
The Daily Mail posted a news article about Manchester United’s Darren Fletcher, who recently underwent his third surgery for UC. Over the last few years, Fletcher has frequently struggled to stay fit. He has played just thirteen games since December 2011.
Multiple surgeries, as in Fletcher’s case, are not uncommon. UC spreads and deeply infects the lining of a patient’s colon and rectum. Although there is no known cure, correctly applied therapy has been known to markedly reduce symptoms and even lead to long-term remission.
Herman & Wallace offered their first on Bowel Pathology and Function in Stony Brook, NY last month and is in the midst of confirming dates for another course in 2014. Keep a look out for updates!