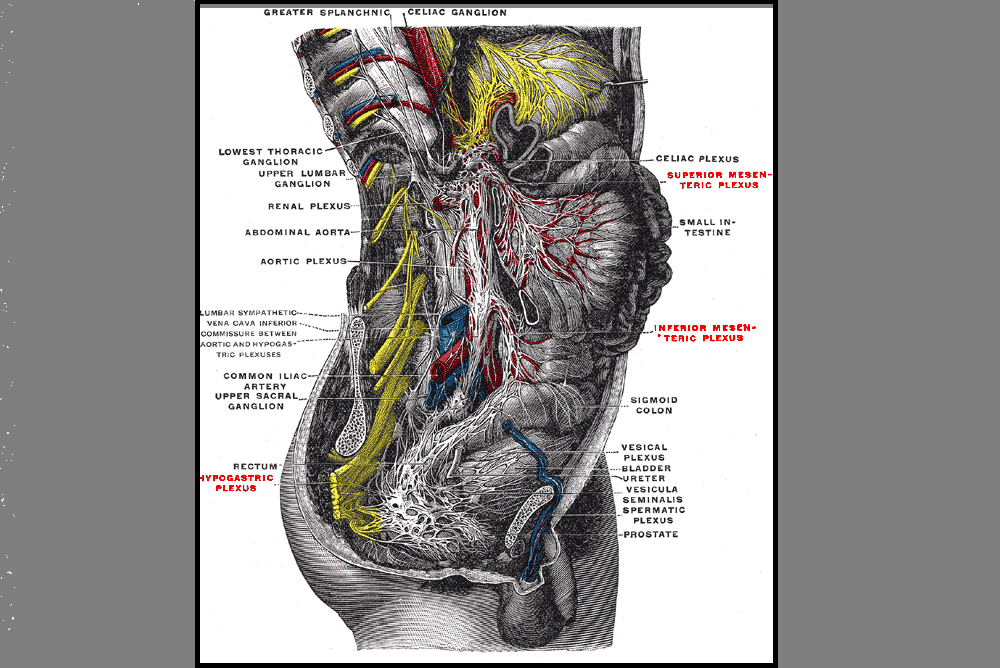
Pelvic health physical therapy sits at the intersection of multiple body systems: musculoskeletal, neuromuscular, gastrointestinal, urologic, reproductive, and psychological. Yet one critical piece is consistently underemphasized in clinical training: pharmacology.
As pelvic health providers, we routinely treat patients who are taking medications that directly influence bladder function, bowel motility, hormonal balance, tissue integrity, pain perception, sexual function, and autonomic regulation. If we are not confident in our understanding of those medications, we risk missing key contributors to our patients' symptoms — or worse, misinterpreting clinical presentation altogether.
Expanding pharmacologic literacy for pelvic health providers isn't optional anymore. It's essential.
Medications Influence the Pelvic Floor More Than We Think
Consider how commonly our patients are prescribed medications like these:
- Anticholinergics for overactive bladder
- Beta-3 agonists for urinary urgency
- Hormonal contraceptives or menopausal hormone therapy
- SSRIs and SNRIs
- Muscle relaxants
- Opioids
- Laxatives or stool softeners
Each of these medications can alter tissue quality, muscle tone, coordination, libido, arousal, bowel patterns, or pain processing. For example, oxybutynin (an anticholinergic) may reduce bladder urgency but contribute to constipation — which in turn increases pelvic floor strain. Hormonal changes driven by oral contraceptives or menopause can affect collagen integrity, vaginal tissue health, and load tolerance. Antidepressants may improve mood while simultaneously influencing sexual function or arousal.
When we assess biomechanics without considering pharmacology for pelvic health, we are seeing only part of the picture.
Medication Side Effects Can Mimic or Exacerbate Dysfunction
Patients frequently present with symptoms such as:
- Constipation or bowel irregularity
- Urinary retention, urgency, or frequency
- Sexual dysfunction or decreased arousal
- Vaginal dryness or tissue irritation
- Fatigue or dizziness affecting exercise tolerance
How often are these attributed solely to pelvic floor dysfunction when medication side effects may be a primary contributor? Understanding pharmacodynamics and pharmacokinetics allows us to recognize red flags early, identify medication-induced symptoms, modify exercise dosing appropriately, and collaborate more effectively with prescribing providers. This elevates our clinical reasoning from symptom management to genuinely comprehensive pelvic health care.
Interdisciplinary Collaboration Starts with Pharmacologic Fluency
Pelvic health PTs frequently collaborate with OB-GYNs, urogynecologists, urologists, gastroenterologists, pain specialists, and primary care providers. When we understand the indications for common pelvic medications — their mechanisms of action, contraindications, and side effect profiles — we can communicate clearly, advocate effectively for our patients, and participate meaningfully in care decisions.
This isn't about prescribing. It's about being an informed provider within a multidisciplinary team. Pharmacologic literacy is what allows pelvic health providers to show up at that table as true clinical partners.
Meet the Faculty: Kristina Koch, PT, DPT, CLT, PCES
Kristina Koch, PT, DPT, CLT, PCES is a board-certified specialist in women's health physical therapy and the creator of this course. With over two decades of clinical experience treating pelvic floor dysfunction across all genders and ages, Kristina brings unparalleled depth to this subject. She earned her doctorate from The College of St. Scholastica in 2021 and currently practices in Colorado Springs, CO, where she also serves as a guest lecturer for graduate PT students at Regis University in Denver and provides educational sessions for medical providers and community groups. Kristina has developed this course because she believes pharmacologic literacy is a professional responsibility — not just a clinical nice-to-have.
Invest in the Provider You're Becoming
Pelvic health is evolving rapidly. Our patients are complex. Our role is expanding. To practice at a high level, we must think beyond muscles and manual therapy — we must understand the biochemical and systemic influences that shape pelvic function, including the medications our patients take every day.
If you are ready to deepen your expertise, improve patient outcomes, and elevate your confidence in pharmacology for pelvic health, this is the course you've been waiting for.
Pharmacologic Considerations for the Pelvic Health Provider
Remote Course — Saturday, April 4, 2026 | 8:00 AM Pacific | Live via Zoom
This one-day remote course is designed specifically for pelvic health rehabilitation professionals and covers:
- Urologic medications and their effects on pelvic floor function
- Gastrointestinal pharmacology
- Hormonal therapies including gender-affirming care medications
- Pain medications and their impact on neuromuscular function
- Psychotropic medications and pelvic health
- Medication side effects that influence pelvic function
- Clinical decision-making and interdisciplinary communication
- Non-medication treatment alternatives
Your patients deserve comprehensive care, and you deserve the knowledge to deliver it. Register today at hermanwallace.com — spots are limited.
Pelvic floor rehabilitation plays an important role in helping men prepare for and recover from treatment for prostate and other pelvic cancers. Targeted pelvic rehabilitation interventions can address urinary continence, bowel function, pelvic pain, and sexual health during both preoperative preparation and post treatment recovery.
Men undergoing treatment for pelvic cancers, including prostate, bladder, penile, and testicular cancers, frequently experience changes in pelvic floor function that affect urinary, bowel, and sexual health. Surgical procedures such as radical prostatectomy, radiation therapy, chemotherapy, and reconstructive surgeries can alter pelvic floor muscle coordination, nerve signaling, and connective tissue mobility. As survivorship improves, clinicians are increasingly recognizing the role of pelvic rehabilitation in addressing these functional changes.
Pelvic floor therapy can be introduced both before treatment and during the recovery period. Preoperative pelvic floor training, often referred to as pelvic floor prehabilitation, allows patients to learn correct muscle activation and coordination prior to surgery. For individuals undergoing prostatectomy, this preparation may support earlier return of urinary continence and improve adherence to postoperative rehabilitation programs.
Following cancer treatment, patients may present with a range of pelvic health concerns including urinary incontinence, bowel dysfunction, pelvic pain, sexual dysfunction, and reduced physical confidence. In clinical practice, these symptoms often overlap and may be influenced by muscle weakness, impaired coordination, neural disruption, and tissue changes such as fibrosis.
For pelvic health clinicians, understanding the role of rehabilitation across the cancer care continuum is essential. Targeted pelvic floor interventions can support recovery of bladder control, bowel regulation, and sexual function while also addressing movement patterns, scar tissue mobility, and patient education during survivorship.
Pelvic Floor Prehabilitation Before Prostate Surgery
Preoperative pelvic floor muscle training is increasingly recommended for patients preparing to undergo radical prostatectomy. The goal of prehabilitation is to help patients identify and properly activate the pelvic floor muscles prior to surgery.
In clinical practice, many patients struggle to isolate the pelvic floor muscles without guided instruction. Teaching correct muscle activation before surgery allows patients to develop familiarity with the exercises that will be used during postoperative rehabilitation. This preparation may also improve adherence to home exercise programs during the recovery phase.
Evidence suggests that patients who perform pelvic floor muscle training prior to prostate surgery may experience faster recovery of urinary continence compared with those who begin training only after surgery. Prehabilitation also provides an opportunity to educate patients about expected postoperative changes, bladder management strategies, and activity progression during early recovery.
For many patients, this early education helps reduce anxiety and allows them to approach surgery with a clearer understanding of the rehabilitation process.
The Role of Pelvic Floor Rehabilitation After Treatment
Pelvic rehabilitation is widely recognized as an important component of recovery following prostate cancer treatment. Structured pelvic floor muscle training programs have consistently demonstrated improvements in urinary continence after radical prostatectomy.
Programs that include supervision by a pelvic health clinician often produce stronger outcomes than unsupervised exercise programs. In addition to strengthening the pelvic floor muscles, rehabilitation may address motor control, endurance, and coordination required for functional bladder support.
Accurate muscle activation is an important part of successful rehabilitation. Techniques such as biofeedback, external or internal palpation, and clinician guided instruction can help ensure that patients are engaging the pelvic floor muscles effectively during training.
Radiation therapy directed toward the pelvis can also contribute to long term changes in pelvic floor muscle function. Patients who receive pelvic radiation may develop reduced muscle endurance, altered neuromuscular coordination, and tissue changes related to fibrosis. These factors may contribute to urinary symptoms, bowel dysfunction, or pelvic pain. Rehabilitation strategies may therefore include interventions focused on muscle retraining, tissue mobility, and symptom management.
Addressing Bowel Dysfunction After Colorectal Cancer Treatment
Pelvic rehabilitation may also play an important role in the management of bowel symptoms following colorectal cancer surgery. Patients who undergo low anterior resection may develop a constellation of symptoms commonly referred to as low anterior resection syndrome. These symptoms may include bowel urgency, stool clustering, and reduced bowel control.
Targeted pelvic floor rehabilitation programs that include strengthening exercises, coordination training, and biofeedback have been shown to improve bowel function and quality of life in this patient population.
From a clinical perspective, improving coordination between pelvic floor muscles and abdominal pressure regulation is often a key component of treatment. Education regarding bowel habits, defecation mechanics, and behavioral strategies may also support symptom management.
Supporting Sexual Function During Recovery
Sexual health is another domain in which pelvic rehabilitation can contribute to recovery following pelvic cancer treatment. Pelvic floor muscle training has been shown to support improvements in erectile function, particularly when incorporated into structured rehabilitation programs before or after prostate surgery.
In addition to strengthening muscles involved in erectile function, pelvic rehabilitation may address pelvic pain, scar tissue sensitivity, and muscle coordination impairments that can contribute to discomfort during sexual activity.
Because sexual health concerns often involve both physical and psychosocial components, collaboration with urology, sexual medicine, and mental health professionals can provide comprehensive support for patients navigating these changes during survivorship.
The Expanding Role of Pelvic Rehabilitation in Cancer Survivorship
As survival rates continue to improve for men with pelvic cancers, greater attention is being placed on functional recovery and quality of life following treatment. Pelvic rehabilitation provides clinicians with an opportunity to address bladder control, bowel function, sexual health, and pelvic pain in a structured and evidence informed way.
For many patients, these symptoms are not simply unavoidable consequences of cancer treatment. With appropriate rehabilitation strategies and coordinated care, meaningful improvements in daily function and overall wellbeing are often achievable.
Clinical Takeaways for Pelvic Health Clinicians
Pelvic rehabilitation plays an important role in supporting functional recovery for male pelvic cancer survivors. Clinicians working with this population may consider the following points.
Urinary incontinence is common following prostatectomy, and pelvic floor muscle training can support recovery of bladder control.
Preoperative pelvic floor training can help patients identify and activate the pelvic floor muscles prior to surgery, which may facilitate postoperative rehabilitation.
Radiation therapy may contribute to changes in pelvic floor muscle endurance, neuromuscular coordination, and connective tissue mobility that influence pelvic function.
Patients undergoing colorectal cancer surgery may develop bowel dysfunction that can respond to pelvic floor rehabilitation interventions.
Pelvic floor muscle training may also support recovery of erectile function and improve sexual health outcomes following prostate cancer treatment.
Collaboration among pelvic health clinicians, urologists, oncologists, and other specialists can improve patient outcomes during cancer survivorship.
Frequently Asked Questions About Pelvic Rehabilitation After Pelvic Cancer
What is pelvic floor therapy after prostate surgery?
Pelvic floor therapy focuses on restoring bladder control, improving pelvic floor muscle coordination, and supporting recovery of sexual function following prostate surgery.
When should pelvic floor therapy begin for prostate cancer patients?
Pelvic floor muscle training may begin prior to surgery as part of prehabilitation and continue after surgery during recovery.
Can pelvic rehabilitation support erectile function recovery?
Pelvic floor muscle training may support erectile function by improving muscle strength and coordination involved in sexual performance.
Is pelvic rehabilitation helpful for bowel dysfunction after colorectal cancer surgery?
Yes. Pelvic rehabilitation can help improve bowel control and coordination for patients experiencing symptoms associated with low anterior resection syndrome.
Who may benefit from pelvic cancer survivorship rehabilitation?
Men treated for prostate, bladder, colorectal, penile, or testicular cancers may benefit from pelvic rehabilitation to address bladder, bowel, sexual, and pelvic pain concerns.
Key Research References
Jones H, et al. Rehabilitation strategies for low anterior resection syndrome following colorectal cancer surgery. Colorectal Dis. 2024.
Kim HJ, Oh SY. Pelvic floor rehabilitation for bowel dysfunction after colorectal cancer surgery. Ann Coloproctol. 2023.
Anderson CA, Omar MI, Campbell SE, et al. Conservative management for postprostatectomy urinary incontinence. Cochrane Database Syst Rev. 2015.
Fernández RA, et al. Pelvic floor muscle training after radical prostatectomy improves urinary continence recovery. Urology. 2015.
Ribeiro LH, et al. Pelvic floor muscle morphology and function after pelvic radiation therapy. Neurourol Urodyn. 2021.
Centemero A, Rigatti L, Giraudo D, et al. Preoperative pelvic floor muscle exercise for early continence after radical prostatectomy. Eur Urol. 2010.
Milios JE, Ackland TR, Green DJ. Pelvic floor muscle training in radical prostatectomy. BMC Urol. 2019.
Dorey G, Speakman M, Feneley R, et al. Pelvic floor exercises for erectile dysfunction. BJU Int. 2005.
Want to learn more about Oncology of the Pelvic Floor?
Recommended Courses: Oncology of the Pelvic Floor Level 1 & Oncology of the Pelvic Floor Level 2A
"Oncology of the Pelvic Floor Level 2A" with Nicole L Dugan,PT,DPT,MSOD, CLT-LANA, WCS
Upcoming Remote Course: April 18-19, 2026 Oncology of the Pelvic Floor Level 2A
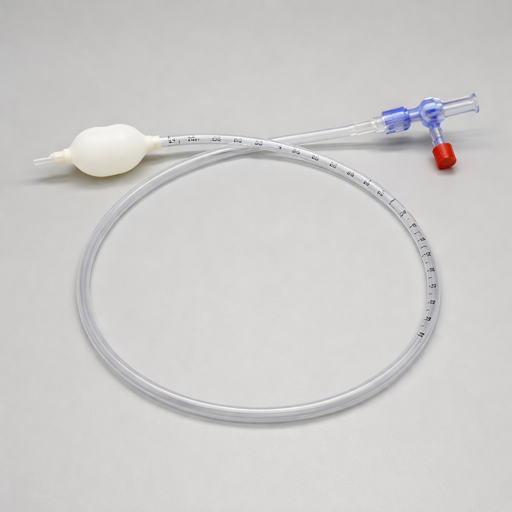
Anorectal balloon catheter training is one of the most underrated but helpful treatments for people with pelvic floor symptoms related to bowel dysfunction. This is a tool that many clinicians don’t know about or are afraid to initiate with their clients. Clinicians wonder if clients will be receptive, how to use an anorectal balloon catheter efficiently, and frequently wonder what cases are appropriate for this specific modality. Anorectal balloon catheter training is a versatile treatment helping patients with pelvic floor conditions that stem from hyposensitivity or hypersensitivity in the rectal canal.
Rehab clinicians can use anorectal balloon catheters to help with defecation training, anorectal sensory training, coordination training, and resistance training that can improve symptoms for individuals with fecal incontinence, fecal urgency, and chronic constipation as well as other colorectal diagnoses. This modality can be used to improve the coordination between the pelvic floor muscles and the abdominal muscles to assist in defecation training. It also can help a patient learn what the urge to have a bowel movement should feel like, especially if they have altered sensation in the anal canal.
An anorectal balloon is a form of biofeedback to use with pelvic floor patients. During treatment, an anorectal balloon is placed in the rectal canal. The balloon can hold 400 mL but filling volumes are typically much lower. The balloon is then filled with air and the amount of air is altered in order to help retrain sensation in the anorectal area. Before implementing this treatment technique in a patient’s plan of care, there are a few steps a rehabilitation provider should take.
First, patients should be screened to make sure they are good candidates for this treatment. This includes internal muscle assessment of the rectal canal prior to implementing training. Detailed patient education on the purpose and procedure of training with an anorectal balloon catheter should be provided. Patients may have some experience with anorectal manometry and may need their therapist to differentiate how manometry testing is for assessment purposes, but balloon training is a biofeedback tool.
Once this treatment is decided upon, the therapist will begin by getting some baseline measurements. These include the first feeling of sensation of the balloon filling, the first urge to defecate, and then their maximum tolerance. These baselines give a provider information on how to proceed with treatment. It is helpful to have norms readily available to be able to compare your patient’s readings to. Caution should be taken when working with patients who have had lower bowel surgeries and pediatric patients, avoiding maximum values beyond a certain value.
With proper consideration of the baseline measurements of sensation levels, a treatment plan can be developed with the use of anorectal balloon training to improve sensation and awareness in the anorectal area. Sensation is trained via inflations and deflations of the balloon to assist in feedback to allow the patient to recognize what normal range values feel like.
Anorectal Balloon Catheters - Intro and Practical Application is a mini-course offered by Herman & Wallace to help providers feel comfortable screening patients for their eligibility for this intervention. The course will assist in helping practitioners to feel confident in providing this treatment with appropriate patients. This class is built with treatment in mind, and intended for therapists who have some exposure to the concept of anorectal assessment and treatment but want to learn more ways to apply this technique to their clients. This class includes didactic information and hands-on lab practice in the privacy of participant’s own space, to help bring this skill to their clinical practice. The next offering of this course is:
https://hermanwallace.com/continuing-education-courses/anorectal-balloon-catheters/

Abdominal bloating and distension are common symptoms reported in pelvic health practice. While many individuals experience occasional bloating that resolves without intervention, persistent or long-standing distension can significantly impact quality of life. Patients often report discomfort, sleep disruption, dietary restrictions, and frustration when symptoms persist without clear answers.
One condition associated with these symptoms is abdomino-phrenic dyssynergia, a disorder involving a paradoxical relationship between the diaphragm and abdominal wall.
Under normal conditions, when intraluminal gas increases in the gastrointestinal tract, the body responds with a coordinated pattern:
- the diaphragm relaxes, and
- the abdominal wall contracts
This response helps maintain abdominal shape and pressure regulation.
However, in abdomino-phrenic dyssynergia, the opposite pattern occurs. The diaphragm contracts downward while the abdominal musculature relaxes, leading to visible abdominal distension and discomfort. Dysfunction of the pelvic floor is also frequently associated with this condition, reinforcing the importance of a comprehensive evaluation of the entire pressure management system.
Traditional management strategies include biofeedback therapy and breathing retraining, both aimed at restoring appropriate neuromuscular coordination.
A Clinical Case Example
In our clinic, we are seeing an increasing number of referrals for patients diagnosed with abdomino-phrenic dyssynergia. One recent patient illustrates how breathing mechanics and musculoskeletal restrictions can contribute to these symptoms.
The patient was a 72-year-old female with a long-standing history of abdominal bloating and distension.
She reported:
- Bloating and abdominal distension throughout the day, worsening toward evening
- Limiting evening food intake due to abdominal discomfort and “tightness”
- Pain rated 3–5/10 in the morning, increasing to 8/10 by late evening
- Difficulty sleeping due to the abdomen feeling “hard and tight” at bedtime
Examination Findings
Physical examination revealed several contributing factors:
- Significant tightness in the posterior chain and erector spinae in the thoracic and lumbar regions
- Reduced thoracic rotation and mobility
- Connective tissue restrictions in the upper abdominal quadrants, especially the epigastric region and inferior rib cage
- Decreased lower rib cage mobility
- Difficulty producing a prolonged or forceful exhale
- Reduced ability to relax the pelvic floor following contraction
These findings highlighted the interaction between breathing mechanics, rib cage mobility, myofascial restrictions, and pelvic floor coordination.
Treatment Approach
Treatment included a multi-system approach addressing breathing, mobility, and neuromuscular coordination.
Interventions included:
- Biofeedback therapy
- Visceral mobilization techniques
- Thoracic spine and rib joint mobilizations
- Soft tissue techniques, including gentle diaphragm release
- Breathing retraining
- Techniques focused on pelvic floor relaxation
The patient completed nine treatment sessions, combined with a structured home maintenance program that she followed consistently.
Outcomes
By the end of treatment, the patient reported:
- 70% overall improvement in symptoms
- Ability to eat evening meals without discomfort
- Restful sleep through the night without abdominal tightness
This case highlights how restoring efficient breathing mechanics and rib cage mobility can significantly influence abdominal pressure regulation, pelvic floor function, and patient comfort.
Why Breathing Matters for Pelvic and Orthopedic Therapists
Breathing is far more than a respiratory function. The diaphragm plays a central role in:
- pressure regulation
- core stability
- pelvic floor coordination
- movement efficiency
Understanding how breathing integrates with the musculoskeletal system can significantly expand a clinician’s ability to address persistent symptoms that may otherwise be overlooked.
In the course Breathing and the Diaphragm: Pelvic and Orthopedic Therapists, we explore these relationships in depth and provide clinicians with practical tools to assess and treat dysfunctional breathing patterns.
Participants will learn how to:
- Explain normal diaphragmatic breathing and the role of the internal and external oblique musculature
- Assess and treat dysfunctional breathing patterns including chest, abdominal, and paradoxical breathing
- Understand the role of intra-abdominal pressure (IAP) in spinal stability
- Apply the concept of regional interdependence in patients with pelvic or back pain
- Recognize how postural patterns influence diaphragm and pelvic floor function
- Identify myofascial contributors to dysfunctional breathing and apply appropriate treatment techniques
- Perform rib and thoracic spine mobilizations to improve respiratory mechanics
- Develop exercise progressions for breathing retraining in clinic and home programs
- Integrate diaphragmatic breathing strategies into athletic rehabilitation
Understanding the relationship between breathing mechanics, mobility, and pelvic floor function allows clinicians to address dysfunction from a more integrated perspective and can lead to meaningful improvements in patient outcomes.
Aparna Rajagopal, PT, MHS, WCS, PRPC, Capp-OB Certified is the lead therapist at Henry Ford Macomb Hospital's pelvic dysfunction program, where she treats pelvic rehab patients and consults with the sports therapy team. Her interest in treating peripartum patients and athletes allowed her to recognize the role that breathing plays in pelvic dysfunction.
Leeann Taptich DPT, SCS, MTC, CSCS leads the Sports Physical Therapy team at Henry Ford Macomb Hospital where she mentors a team of therapists. She also works very closely with the pelvic team at the hospital which gives her a very unique perspective of the athlete.
Aparna and Leeann co-authored the course, Breathing and the Diaphragm: Pelvic and Orthopedic Therapists, which helps clinicians understand breathing mechanics and their relationship to the pelvic floor.
Course Dates: March 14, 2026
Price: $450
Experience Level: Beginner
Contact Hours: 13.5
Description: This remote course is an integrated approach where participants will learn how the diaphragm, breathing, and the abdominals can affect core and postural stability through intra-abdominal pressure changes while looking at structures from the glottis and the cervical region to the pelvic floor.
This course includes assessment and treatment of the barriers by addressing thoracic spine articulation and rib cage abnormalities in the fascial system of muscles related to breathing and the diaphragm. Instructed techniques are applicable to patients who present with Diastasis Rectus Abdominis, pelvic pain, incontinence, and prolapse, as well as cervical, thoracic, scapular, and lumbar pain.

I recently evaluated a 75 y.o patient who presented with significant urinary urgency and frequency, voiding approximately every hour. She reported disrupted sleep due to nocturia, stating, “I can’t sleep at night because I keep getting up to go to the bathroom. They gave me medication to help me sleep, but it doesn’t work.”
Over the course of the visit, it became clear that she was also experiencing chronic anxiety. Anxiety permeated multiple aspects of her daily life, she worried about day-to-day events as well as events in the future. She reported that her urinary symptoms worsened during periods of heightened anxiety, and she had difficulty relaxing both her body and mind.
My initial clinical focus was nervous system regulation. I guided her to sit back comfortably and take several gentle breaths, emphasizing a prolonged exhalation with an audible sigh. She was instructed to consciously release tension throughout her body while maintaining attention on her breath. After only a few breaths, she smiled and reported that she already felt calmer.
In addition to a home program that included diaphragmatic breathing, self–abdominal massage, and pelvic girdle mobility exercises, I introduced two Acupressure points for nervous system self-regulation: Conception Vessel 17 (CV17) and Yintang (EX-HN 3).
CV17, located at the center of the chest, is traditionally associated with emotional regulation and calming of the heart-mind connection. Yintang, located between the eyebrows, is described in Traditional Chinese Medicine (TCM) as having a mentally stabilizing and calming effect.¹
At her subsequent visit, the patient reported feeling calmer overall and noted that she was able to use the Acupressure points independently to regulate her anxiety. Over the course of several visits, an integrative plan addressing hip mobility, bladder training, behavioral modification and nervous system regulation resulted in measurable improvement. Her daytime voiding interval increased to approximately 2.5 hours, and nocturnal voiding frequency also decreased.
Acupressure as an Evidence-Informed Integrative practice
Acupressure, rooted in Traditional Chinese Medicine, is increasingly recognized as an evidence-informed, integrative, and trauma-informed intervention. Integrative health and medicine approaches intentionally combine conventional physical therapy interventions with holistic strategies that address the whole person - physically, mentally, emotionally, and spiritually (Justice et al).
The use of Acupressure for anxiety is well established in integrative medicine. Acupoints such as Yin Tang (EX-HN3), Shenmen (HT7), Neiguan (P6), Hegu (LI4), Taichong (LV3), Jianjing (GB21), Zu San Li (ST36) and Sanyinjiao (SP6) are some of the most frequently used points to treat anxiety2. Yintang (EX-HN 3), in particular, has demonstrated anxiolytic effects and has also been associated with improvements in depressive symptoms.³
Beyond mental health applications, Acupressure has also been used as an effective non-pharmacological therapy for the management of a host of conditions such as insomnia, chronic pelvic pain, dysmenorrhea, infertility, constipation, digestive disorders and urinary dysfunctions. Emerging research suggests that Acupressure influences neural networks across multiple systems, supporting emotional regulation and multisystem healing
Physiologically, Acupressure has been shown to improve heart rate variability and reduce sympathetic nervous system activity. This downregulation is associated with decreased release of stress hormones such as epinephrine and cortisol, facilitating the relaxation response and correlating with reductions in anxiety and pain.
Why Acupressure Matters in Pelvic Health Rehabilitation
The pelvic floor is highly responsive to stress, anxiety, and unresolved trauma, often demonstrating increased tone or guarding in response to perceived threat. This can contribute to pelvic pain, urinary dysfunction, dyspareunia, constipation, and other pelvic health conditions.
These presentations are not purely musculoskeletal, they frequently reflect underlying nervous system dysregulation. Incorporating Acupressure into pelvic health rehabilitation can meaningfully support patients by:
· Calming hyperactive pelvic and autonomic nerves
· Improving circulation and tissue mobility in the pelvic region
· Releasing stored muscular tension and trauma
· Supporting emotional grounding, safety, and resilience
Acupressure can be particularly beneficial during or after pregnancy, childbirth, surgery, or emotionally traumatic experiences, offering a gentle, patient-empowering approach to healing.
Acupressure as a Hands-On Self-Regulation Tool
Acupressure involves the application of gentle, intentional pressure to specific points along the body’s meridian system. These points correspond with key organ systems, including the nervous, digestive, and reproductive systems and can influence both physical and emotional health.
Clinical benefits of acupressure include:
· Vagal nerve modulation and stress reduction
· Decreased muscle tension and chronic pain
· Enhanced emotional regulation and trauma support
· Promotion of relaxation and improved sleep
Integrating acupressure into pelvic health physical therapy supports whole-person healing, restoring not only movement and function, but also a sense of safety, stability, and emotional balance.
Commonly Used Acupressure Points for Anxiety, Pain, and Pelvic Health
· CV 17 (Conception Vessel 17) – Located at the center of the chest Main point for Emotional healing
· Yintang (EX-HN 3) – Located between the eyebrows Mentally stabilizing effect, calming point
· H 7 ( Heart 7) – Located on the ulnar side of the hand, in the joint space) Helps with Insomnia, reduces anxiety
· P 6 (Pericardium 6) – Inner forearm Calms the heart, reduces anxiety and nausea
· Sp 6 (Spleen 6) – Above the inner ankle Regulates reproductive health
· CV 6 (Conception Vessel) – Below the navel Supports core energy, fatigue and abdominal tension
These points can be gently stimulated during therapy or taught as part of a home program, offering patients the tools for emotional self-regulation. To explore these concepts further, please join us for the upcoming remote course Acupressure for Optimal Pelvic Health scheduled for Feb 7th & 8th . This course introduces participants to foundational principles of Traditional Chinese Medicine, Acupuncture, and Acupressure, with a focused exploration of the Bladder, Kidney, Stomach, and Spleen meridians.
Participants will also learn additional nervous system–regulating points for managing anxiety, pain, and related symptoms, as well as two comprehensive acupressure-based home and wellness programs. The course further integrates Yin yoga as a complementary practice, offering an evidence-informed perspective on how Yin postures associated with specific meridians may influence neurodynamic pathways and support multidimensional healing.
References
1. Chen SR, Hou WH, Lai JN, Kwong JSW, Lin PC. Effects of Acupressure on Anxiety: A Systematic Review and Meta-Analysis. J Integr Complement Med. 2022;28(1):25-35. doi:10.1089/jicm.2020.0256
2. Yang J, Do A, Mallory MJ, Wahner-Roedler DL, Chon TY, Bauer BA. Acupressure: An Effective and Feasible Alternative Treatment for Anxiety During the COVID-19 Pandemic. Glob Adv Health Med. 2021;10:21649561211058076. Published 2021 Dec 12. doi:10.1177/21649561211058076
3. Kwon CY, Lee B. Acupuncture or Acupressure on Yintang (EX-HN 3) for Anxiety: A Preliminary Review. Med Acupunct. 2018;30(2):73-79. doi:10.1089/acu.2017.1268
4. Justice C, Sullivan MB, Van Demark CB, Davis CM, Erb M. Guiding Principles for the Practice of Integrative Physical Therapy. Phys Ther. 2023;103(12):pzad138. doi:10.1093/ptj/pzad138
5. Monson E, Arney D, Benham B, et al. Beyond Pills: Acupressure Impact on Self-Rated Pain and Anxiety Scores. J Altern Complement Med. 2019;25(5):517-521.
6. Abaraogu UO, Igwe SE, Tabansi-Ochiogu CS. Effectiveness of SP6 (Sanyinjiao) acupressure for relief of primary dysmenorrhea symptoms: A systematic review with meta- and sensitivity analyses. Complement Ther Clin Pract. 2016;25:92-105
7. He Y, Guo X, May BH, et al. Clinical Evidence for Association of Acupuncture and Acupressure With Improved Cancer Pain: A Systematic Review and Meta-Analysis. JAMA Oncol. 2020;6(2):271-278. doi:10.1001/jamaoncol.2019.5233
8. Hasanin ME, Elsayed SH, Taha MM. Effect of Acupressure on Anxiety and Pain Levels in Primiparous Women During Normal Labor: A Randomized Controlled Trial. J Integr Complement Med. 2024;30(7):654-661. doi:10.1089/jicm.2023.0072

Pain shows up in almost every pelvic health plan of care, but a lot of providers were never actually trained to teach pain in a way that changes outcomes. We learn how to assess tissues, strength, tone, and pathology, but pain is not a simple “damage meter.” It’s a protective output of the nervous system, shaped by context, perceived threat, inflammation, prior experiences, hormones, and learned responses. When we skip pain science education (or keep it vague), patients often stay fearful, hypervigilant, and stuck, especially when imaging is normal or symptoms don’t “match” what we see.
Endometriosis, among other pelvic pain conditions, is one of the clearest examples of why this matters. Endometriosis is characterized by lesions containing endometrium-like epithelium and stroma that develop outside the uterus and are biologically distinct from normal uterine endometrium. Endo is a real inflammatory disease with lesions that can be found on the bowel, bladder, ureters, abdominal wall, and peritoneum commonly. And yet pain severity doesn’t reliably correlate with lesion size, number, or location. Some patients with extensive disease report minimal pain, while others with smaller disease experience life-altering symptoms. Pain science helps us explain that gap: the nervous system can become sensitized over time, turning up the volume on danger signals even when tissues are stable or after the primary driver has been addressed.
Clinically, one of the most important skills is being able to distinguish peripheral pain generators from sensitization. Peripheral drivers include things like active lesions/inflammation, adhesions, pelvic floor overactivity, tissue irritation, and organ-specific contributors. Sensitization shows up when pain persists beyond expected healing, spreads, becomes disproportionate to findings, or is paired with hypervigilance, fear-avoidance, and strong symptom reactivity to stress, sleep disruption, and attention. In pelvic health, cross-talk between organs adds another layer, bladder, bowel, uterus, and pelvic floor can share neural pathways, so symptoms don’t always point neatly to the true source.
This is where pain science education becomes a part of treatment. For endometriosis, an example of pain science education starting point could be: “Endo can absolutely create pain through inflammation and lesion activity, but pain isn’t always a direct reflection of how much disease is present. Over time, your nervous system can become extra protective, like an alarm system that’s gotten too sensitive. That doesn’t mean the pain is in your head. It means your pain IS real, and your nervous system is amplifying signals. The good news is the system can be retrained through the right combination of medical care, pelvic rehab, graded exposure, and nervous system regulation.”
When providers can explain pain clearly, patients stop interpreting every symptom spike as damage. They become more confident with movement, more consistent with rehab, and more resilient during flares. And that’s why pain science education is so important, because with endometriosis, IC/PBS, prostatitis, IBS, vaginismus/dyspareunia, and primary dysmenorrhea, your hands matter, but what you do with your words can be the turning point.
If you’re treating endometriosis, or any chronic pelvic pain condition, and you’re not sure whether you’re addressing the “spark” (peripheral drivers) versus the “fuel” (sensitization), this Pain Science class is designed to make that clinical reasoning practical, teachable, and immediately usable in your sessions.
Dr. Tara Sullivan, PT, DPT, PRPC, WCS, IF Sexual Medicine in Pelvic Rehabilitation - Remote Course - March 14-15 2026
Learn More: Sexual Medicine in Pelvic Rehab March 14-15, 2026

When we think of sports rehab, we typically envision athletes returning to the court after an ankle sprain or knee injury. But what if the same principles of rigorous assessment, load transfer optimization, movement education, and functional stability could apply to one of the body’s most critical yet under‑appreciated joints: the sacroiliac joint (SIJ)?
For clinicians working in pelvic health, embracing a sports‑rehab mindset can transform how we evaluate and treat SIJ dysfunction and pain, and recent research supports this crossover approach. Now might just be the ideal time to integrate these strategies into your practice.
Why Sports Rehab Principles Matter for SIJ/Pelvic Health
- High prevalence of SIJ issues in athletic populations: A 2024 systematic review found that among athletes, average prevalence of SIJP or SIJD was ~10.7%, with rates much higher (32–36%) in those presenting with low back or pelvic pain. (Mirdamadi et al, 2025)
- SIJ dysfunction often coexists with lower extremity injuries: In a 2023 study of basketball players, those with SIJ pain or dysfunction reported significantly more lower-limb and pelvic‑girdle injuries, both acute and overuse, than their peers without SIJ complaints. (Abdollahi et al, 2023)
- Biomechanics and load transfer matter: The SIJ plays a key role in transmitting loads between the spine and lower extremities. Disruption in force transmission, whether from muscular imbalance, altered movement patterns, or pelvic instability, may contribute not just to SIJ pain, but to secondary injuries elsewhere. (Prather, 2000; Abdollahi et al, 2023)
These findings align closely with core sports‑rehab principles: assessing mechanical and neuromuscular impairments, correct faulty movement or load patterns, and restore stability and function before returning to high‑demand activity.
Translating Sports‑Rehab Strategies into Pelvic Health Practice
Here are some of the evidence-based crossover strategies that pelvic rehab clinicians can begin using:
- Comprehensive Functional Assessment: Rather than isolating the SIJ, consider the entire kinetic chain. For instance, when evaluating an athlete (or active individual) with lower extremity complaints, include SIJ screening - given its association with lower‑limb injuries. (Abdollahi et al, 2023)
- Dynamic & Stabilization Exercises: Inspired by sports rehab protocols, incorporating exercises that challenge stability, proprioception, and dynamic load transfer can be effective. A case study combining Swiss‑ball training, mobilization with movement, and taping demonstrated promising results in an athlete with concurrent SIJD and ankle sprain. (Shedge et al, 2024)
- Manual Therapy + Movement Integration: While manual therapy alone has mixed evidence, a recent meta‑analysis found that SIJ manual therapy did not significantly reduce pain vs. non-manual interventions but did improve disability moderately. Combining manual therapy with functional movement and exercise tends to align with best-practice sports rehab philosophy. (Trager at al, 2024)
- Holistic, Movement‑Based Rehab Over “Fixing” Passive Structures: Rather than focusing solely on static “joint alignment” or joint‑centric correction, prioritize restoring functional movement, load capacity, and neuromuscular control - all principles that athletic trainers and sports PTs rely on routinely.
Why This Matters - For Both Clinicians and Clients
Adapting a sports‑rehab informed paradigm for SIJ/pelvic health offers several advantages:
- Broader applicability: Whether your client is a weekend runner, a postpartum individual, or a competitive athlete, a functional, movement‑based SIJ rehabilitation model supports real‑world demands.
- Better injury prevention and reduced recurrence: By addressing underlying load‑transfer dysfunction and neuromuscular control, not just symptoms, you may reduce the risk of future pelvic or lower‑extremity injuries.
- Improved patient buy-in: Clients often resonate with language around “stability,” “function,” and “return to activity.” Familiar terms from sports rehab, which can increase compliance and perceived relevance.
Connect the Dots Between Sports Rehab & Pelvic Rehab - Take the 4‑Hour Course
If you’re ready to bridge the gap between sports rehab and pelvic health, I invite you to join the upcoming four‑hour remote course, Sacroiliac Joint Current Concepts, taught by experienced former NHL physical therapist and athletic trainer Steve Dischiavi, PT, PhD, DPT, MPT, SCS, ATC, COMT.
📅 Date: January 25, 2026
📚 You’ll receive:
- A full, easy‑to-follow SIJ exam sequence - optimized for a pelvic‑health context.
- Treatment strategies aligned with current evidence and sports rehab biomechanics.
- Practical tools you can integrate into your clinical practice immediately (Monday morning!)
Transform your approach and help clients move, perform, and heal better. Register today to reserve your spot.
References
- Mirdamadi N, Khadembashiri MM, Moghadam N, Kordi R. Prevalence and Risk Factors of Sacroiliac Joint Pain in Athletes: A Systematic Review and Proportional Meta-Analysis. Clin J Sport Med. 2025 Mar 26;35(4):514-525. doi: 10.1097/JSM.0000000000001341. PMID: 40135982.
- Abdollahi, S., Sheikhhoseini, R., Rahimi, M. et al. The sacroiliac dysfunction and pain is associated with history of lower extremity sport related injuries. BMC Sports Sci Med Rehabil 15, 36 (2023). https://doi.org/10.1186/s13102-023-00648-w
- Prather H. Pelvis and sacral dysfunction in sports and exercise. Phys Med Rehabil Clin N Am. 2000 Nov;11(4):805-36, viii. PMID: 11092020.
- Shedge SS, Ramteke SU, Samal S. Integrated Rehabilitation Approach Utilizing Swiss Ball Training, Mulligan Taping, and Mobilization With Movement for Simultaneous Management of Sacroiliac Joint Dysfunction and Lateral Ankle Sprain in a Badminton Athlete: A Case Study. Cureus. 2024 Mar 26;16(3):e56942. doi: 10.7759/cureus.56942. PMID: 38665699; PMCID: PMC11044192.
- Trager RJ, Baumann AN, Rogers H, Tidd J, Orellana K, Preston G, Baldwin K. Efficacy of manual therapy for sacroiliac joint pain syndrome: a systematic review and meta-analysis of randomized controlled trials. J Man Manip Ther. 2024 Dec;32(6):561-572. doi: 10.1080/10669817.2024.2316420. Epub 2024 Feb 14. PMID: 38353102; PMCID: PMC11578406.

Pelvic floor rehabilitation is often associated with urinary continence or pelvic support, but recent research highlights its broader role, including significant impacts on respiratory health. Studies now suggest that combining pelvic floor muscle (PFM) training with pulmonary rehabilitation can enhance lung function, particularly in elderly patients recovering from orthopedic surgery.
Pelvic Floor Muscle Training and Pulmonary Function
A 2025 randomized controlled trial investigated the effects of combining pelvic floor muscle training with pulmonary rehabilitation in elderly patients following surgery for intertrochanteric femur fractures (Ji et al., 2025). Fifty patients were randomly assigned to either pulmonary rehabilitation alone or pulmonary rehabilitation combined with PFM training. After four weeks, both groups showed improvements in forced vital capacity (FVC), peak expiratory flow (PEF), and the FEV1/FVC ratio, with the combined intervention group demonstrating significantly greater gains. Diaphragm excursion and thickening fraction were also improved, suggesting a synergistic relationship between the diaphragm and pelvic floor muscles that enhances respiratory mechanics.
Supporting Evidence
Additional studies support the connection between PFM function and respiratory performance. A recent study using sensor-based diaphragm exercises combined with PFM training in women with stress urinary incontinence demonstrated improvements in both pelvic floor function and respiratory parameters (Yakıt Yeşilyurt et al., 2025). Similarly, pelvic floor electrical stimulation has been shown to enhance diaphragm excursion and rib-cage movement during tidal and forceful breathing and coughing (Hwang et al., 2021). Foundational work also demonstrated that co-activation of abdominal and pelvic floor muscles contributes to improved expiratory function and intra-abdominal pressure regulation (Sapsford et al., 2001). Together, these studies highlight the physiological link between the pelvic floor, diaphragm, and respiratory system.
Why This Matters
For elderly patients recovering from hip fractures, optimizing lung function is critical to reducing postoperative complications such as pneumonia and supporting overall recovery. Integrating PFM training with pulmonary rehabilitation provides a novel and underutilized approach to enhance respiratory efficiency and accelerate functional recovery. Moreover, these findings expand the role of pelvic floor rehabilitation beyond traditional urogenital outcomes, emphasizing its value in multidisciplinary rehabilitation programs.
Takeaway
The pelvic floor contributes significantly to respiratory mechanics. Combining pelvic floor muscle training with pulmonary rehabilitation can improve lung function in elderly post-surgical patients and may support broader recovery goals. As research evolves, pelvic floor specialists have the potential to play a key role in integrated rehabilitation approaches.
Practical Next Step: Elevate your clinical expertise by enrolling in Breathing and the Diaphragm, scheduled for December 6. This course covers diaphragm anatomy, breathing mechanics, and how the diaphragm, abdominals, and pelvic floor interact to regulate intra‑abdominal pressure, support core stability, and influence posture. Lab sessions include assessment and treatment of dysfunctional breathing patterns, ribcage and thoracic-spine restrictions, and practical strategies for clinical integration. While broadly applicable to pelvic pain, incontinence, prolapse, and core/abdominal issues, these techniques can be adapted for elderly post-hip-fracture patients to optimize lung function and recovery.
References
- Yakıt Yeşilyurt S, Şahiner Pıçak G, Başol Göksülük M, Balıkoğlu M, Özengin N. Investigating the Effectiveness of Pelvic Floor Muscle Training, Including Sensor-Based Diaphragm Exercises in Women With Stress Urinary Incontinence: A Randomized Controlled Study. Arch Phys Med Rehabil. 2025 Jul 11:S0003-9993(25)00796-8. doi: 10.1016/j.apmr.2025.06.019. Epub ahead of print. PMID: 40653184. https://pubmed.ncbi.nlm.nih.gov/40653184/
- Ji D, Fu Y, Wu L, Tian C, Jin S. Effect of pelvic floor muscle combined with pulmonary rehabilitation training on lung function in elderly patients after surgery for intertrochanteric fractures of the femur: a randomized controlled trial. Eur J Med Res. 2025 May 14;30(1):381. doi: 10.1186/s40001-025-02610-7. PMID: 40369675; PMCID: PMC12076919. https://pubmed.ncbi.nlm.nih.gov/40369675/
- Hwang UJ, Lee MS, Jung SH, Ahn SH, Kwon OY. Effect of pelvic floor electrical stimulation on diaphragm excursion and rib cage movement during tidal and forceful breathing and coughing in women with stress urinary incontinence: A randomized controlled trial. Medicine (Baltimore). 2021 Jan 8;100(1):e24158. doi: 10.1097/MD.0000000000024158. PMID: 33429797; PMCID: PMC7793445. https://pubmed.ncbi.nlm.nih.gov/33429797/
- Sapsford RR, Hodges PW, Richardson CA, Cooper DH, Markwell SJ, Jull GA. Co-activation of the abdominal and pelvic floor muscles during voluntary exercises. Neurourol Urodyn. 2001;20(1):31-42. doi: 10.1002/1520-6777(2001)20:1<31::aid-nau5>3.0.co;2-p. PMID: 11135380. https://pubmed.ncbi.nlm.nih.gov/11135380/

As the holiday season approaches, Thanksgiving reminds us to pause and reflect on what we are grateful for in our personal lives and in our professional practice. For pelvic health practitioners, one of the greatest sources of gratitude is the opportunity to guide patients toward meaningful progress in their rehabilitation journeys.
Recognizing Small Wins Makes a Big Difference Pelvic rehabilitation is often a journey of incremental improvements. While some changes may be subtle, each step forward is a win. Whether it’s a patient regaining core strength, experiencing reduced pain, improving bladder or bowel control, or building confidence in their body, these victories deserve recognition.
Pelvic rehabilitation is often a journey of incremental improvements. While some changes may be subtle, each step forward is a win. Whether it’s a patient regaining core strength, experiencing reduced pain, improving bladder or bowel control, or building confidence in their body, these victories deserve recognition.
Celebrating small milestones can empower patients, reinforcing that their effort and consistency are yielding real results. Acknowledging progress, even the tiniest, can make a difference in adherence, motivation, and long-term outcomes.
Ways to Celebrate Patient Progress
- Track measurable improvements: Use objective measures like strength, endurance, or symptom diaries to show patients how far they’ve come.
- Highlight functional achievements: Celebrate when a patient reaches a daily-life goal, like returning to exercise, sitting comfortably, or managing symptoms during work or travel.
- Share success stories (with permission): Patient testimonials can motivate others and reinforce a culture of positivity and gratitude in your clinic.
- Verbal acknowledgment: Never underestimate the power of simply saying, “You’ve made great progress!”
Simple Pelvic Floor-Friendly Tips for the Holidays
The holiday season often brings long hours of cooking, hosting, or traveling. Activities that can challenge posture, core stability, and pelvic floor engagement. Here are some short, actionable tips patients can use to stay mindful of their pelvic health:
- Engage the core while standing: While chopping vegetables or stirring a pot, gently draw the belly button toward the spine and lift the pelvic floor to maintain activation.
- Shift weight regularly: Avoid standing in one position for too long. Step side to side or gently march in place while waiting for the oven timer.
- Practice deep breathing: Encourage diaphragmatic breathing while seated or standing to release tension and coordinate with the pelvic floor.
- Take micro-breaks: Every 30–60 minutes, sit or stand with good posture, roll the shoulders, and gently lengthen the spine.
- Travel-friendly stretches: While on the road or sitting for long periods, gently lift and lower the pelvic floor or do seated pelvic tilts to maintain mobility.
 These simple practices can help patients maintain pelvic floor awareness, reduce tension, and feel more comfortable throughout the holiday festivities.
These simple practices can help patients maintain pelvic floor awareness, reduce tension, and feel more comfortable throughout the holiday festivities.
Fostering Gratitude in Your Practice
Showing gratitude to your patients strengthens the therapeutic relationship. A simple thank-you note, a personalized follow-up, or acknowledging their dedication in session can help them feel seen and appreciated. Gratitude flows both ways: as you recognize patients’ efforts, you’re also reminded why you chose this profession - the opportunity to make a meaningful impact in people’s lives.
Looking Ahead with Appreciation
Thanksgiving is a perfect time to reflect on the wins from the past year, both big and small. Take a moment to appreciate the resilience of your patients and the progress you’ve helped facilitate. As we guide patients toward healthier, more empowered lives, celebrating these victories reminds us of the profound value of pelvic rehabilitation work.

Pelvic rehabilitation is a constantly evolving specialty, and your ability to offer high-quality care grows when you have access to a diverse set of tools. Modalities play a significant role in strengthening clinical precision, improving neuromuscular learning, enhancing patient engagement, and expanding treatment possibilities. When you integrate evidence-based modalities into your practice, you elevate the effectiveness and individualization of your patient care.
Here are the top five reasons modalities matter in pelvic rehabilitation, along with examples of tools that can support your work
1. Modalities Improve Your Clinical Precision
Pelvic health presentations often involve complex relationships between muscle tone, coordination, pain, biomechanics, breathing patterns, and emotional factors. Modalities help you see these interactions more clearly and treat them more effectively.
Tools that support clinical precision include:
- Surface EMG biofeedback for assessing activation, resting tone, and timing.
- Real Time Ultrasound Imaging (RUSI) for observing deep core mechanics, pelvic floor movement, and breathing synergy.
- Myofascial tools that help you identify tissue restrictions and monitor tissue response.
- Postural support belts and braces clarify how external stabilization influences symptoms.
With these tools, you gain insights that strengthen your clinical decisions and give patients a clearer understanding of what is happening in their bodies.
2. Modalities Support Both Uptraining and Down Training
Pelvic rehabilitation patients often need help with improving activation, reducing overactivity, coordinating movement, or building endurance. Modalities help you guide the nervous system in the direction that best supports each patient’s goals.
Examples include:
- Surface EMG biofeedback for awareness, facilitation, and relaxation.
- Electrical stimulation to support neuromuscular firing for those with inhibition or weakness.
- Real Time Ultrasound Imaging to help patients visualize diaphragmatic motion and pelvic floor excursion.
- Myofascial tools such as soft tissue instruments to reduce guarding and improve mobility.
- Breath training accessories like visual feedback tools that support better coordination.
Using modalities for both uptraining and down training gives patients more ways to understand and feel the changes you are guiding them toward.
3. Modalities Enhance Patient Understanding and Self-Efficacy
Education and behavior change are central to pelvic rehabilitation. Many patients struggle to conceptualize pelvic floor movement, pressure systems, or muscle relaxation. Modalities make these invisible processes visible and actionable.
Tools that promote self-efficacy include:
- Biofeedback displays that show contraction and relaxation in real time.
- Real-time ultrasound images that reveal how the pelvic floor, transversus abdominis, and diaphragm work together.
- Myofascial release tools that patients can use safely at home.
- Lubricants, dilators, and pelvic wands that support sexual wellness, tissue tolerance, and desensitization.
- Pelvic support belts that help patients feel stable and confident during daily activities.
When patients understand what they are doing and feel empowered by their progress, they become stronger partners in their own recovery.
4. Modalities Expand What Is Possible in Your Treatment Sessions
You already rely on your hands, your knowledge, and your clinical reasoning. Modalities add another layer that allows you to address diverse needs in more targeted ways.
Examples include:
- Myofascial tools for reducing soft tissue restrictions and improving movement.
- Electrical stimulation for supporting continence or reducing pelvic pain.
- Light therapy devices that can help with tissue healing.
- RUSI for assessing pressure management and facilitating motor learning.
- Belts, braces, and supports that can help reduce pain and improve load transfer during movement.
- Lubricants and moisturizers support tissue comfort and sexual function.
Your treatment sessions become more versatile, adaptable, and responsive to the patient in front of you.
5. Modalities Improve Outcomes Across Many Pelvic Health Conditions
The evidence for modality use continues to grow. When you broaden your clinical toolkit, you are better equipped to support patients with varied needs.
Modalities can enhance care for:
- Stress and urge incontinence
- Pelvic pain and overactive pelvic floor conditions
- Bowel dysfunction and dyssynergia
- Dyspareunia and sexual dysfunction
- Postpartum recovery
- Post-prostatectomy rehabilitation
- Vulvodynia and vestibulodynia
- Coordination impairments and breathing dysfunction
- Lumbopelvic instability and load transfer issues
From myofascial support tools to electrical stimulation to imaging and external supports, modalities allow you to tailor interventions with greater specificity and effectiveness.
Build Your Skills with a Hands-On Course Focused on Modalities
If you want clear guidance, supported practice time, and evidence-based instruction on using modalities safely and effectively, Modalities and Pelvic Function was designed for you.
The course combines pre-course video lectures with two days of hands-on lab and dedicated instruction. You will learn how to select and apply modalities, interpret findings, support neuromuscular learning, and integrate tools such as biofeedback, electrical stimulation, myofascial instruments, RUSI, and patient support devices.
Join the Upcoming Boston Course
Venue: Current Medical Technologies
Address: 14 Kendrick Road, Unit 1, Wareham, MA 02571
Dates: January 24 through 25, 2026
Elevate your pelvic rehabilitation practice. Register now to reserve your seat.
By accepting you will be accessing a service provided by a third-party external to https://www.hermanwallace.com/