An 80 year old lady who had seen a physical therapist where I once worked in Naperville, IL, just completed a marathon and a 5k race in one weekend. She is undoubtedly one woman who can change our perception of the “elderly,” but we all know her strength and ability are not the norm. The geriatric patients coming to therapy for pelvic floor disorders are more likely to be too frail to have run a mile this century, and they are most likely struggling with functional ADLs, as research suggests.
 A study by Erekson et al., (2015) looked into the prevalence of frailty, cognitive impairment, and functional disability among women over 65 years of age looking for the best treatment for their pelvic floor dysfunction (PFD). A major concern was the presence of frailty being equated with poorer surgical outcomes. The 150 women in the study were tested with the Fried Frailty Index to measure frailty, the Saint Louis University Mental Status Score for cognitive screening, and the Katz ADL score for functional status. Pelvic organ prolapse was present in 65.3% women, urinary incontinence in 20.7%, overactive bladder in 9.3%, and anal incontinence in 0.7%. Sixteen percent of women were considered frail and 42% were “prefrail.” Dementia was determined in 21.3% of women, and functional disability in 30.7%. Pelvic floor dysfunction in women with frailty caused a significantly greater life-impact than in normal and pre-frail women. Forty-six percent of the subjects opted for surgery, but only women with functional disability, not impaired cognition nor frailty, were less likely to choose non-surgical intervention. The authors concluded that being able to identify women with PFD with risk factors of frailty, cognitive impairment, and functional disability may help predict the risk of complications before surgery and help encourage behavioral changes and provide the appropriate pre and post-operative care for each woman.
A study by Erekson et al., (2015) looked into the prevalence of frailty, cognitive impairment, and functional disability among women over 65 years of age looking for the best treatment for their pelvic floor dysfunction (PFD). A major concern was the presence of frailty being equated with poorer surgical outcomes. The 150 women in the study were tested with the Fried Frailty Index to measure frailty, the Saint Louis University Mental Status Score for cognitive screening, and the Katz ADL score for functional status. Pelvic organ prolapse was present in 65.3% women, urinary incontinence in 20.7%, overactive bladder in 9.3%, and anal incontinence in 0.7%. Sixteen percent of women were considered frail and 42% were “prefrail.” Dementia was determined in 21.3% of women, and functional disability in 30.7%. Pelvic floor dysfunction in women with frailty caused a significantly greater life-impact than in normal and pre-frail women. Forty-six percent of the subjects opted for surgery, but only women with functional disability, not impaired cognition nor frailty, were less likely to choose non-surgical intervention. The authors concluded that being able to identify women with PFD with risk factors of frailty, cognitive impairment, and functional disability may help predict the risk of complications before surgery and help encourage behavioral changes and provide the appropriate pre and post-operative care for each woman.
Silay et al., (2016) published a review on urinary incontinence (UI) in elderly women, relating its association with other geriatric conditions. Sixty-four females aged 65 and older were evaluated using the Turkish version of the International Consultation on Incontinence Questionnaire-Short Form (ICIQ-SF) to assess UI and quality of life. Activities of daily living (ADL) and instrumental activities of daily living (IADL) were used to evaluate functional status, and the Mini Mental State Examination was used for cognitive assessment. The comorbidities, pharmaceuticals, falls, and body mass index (BMI) of patients were also recorded. Results showed the subjects’ rate of urinary incontinence was 40.6%, and 28.1% of the women had their quality of life impacted. There was a statistically significant association using logistic regression between UI and quality of life, functional status, and comorbidity. Sadly, 50% of patients thought UI was normal with aging, 34.6% had been embarrassed to tell anyone about it, and 15.3% said they did not know UI was something for which medical treatment could be given.
Understanding how to manage frailty, cognitive issues, and functional deficits of our elderly patients can positively impact treatment outcomes. We should always strive to educate our patients and be aware of conditions that may be affecting or even contributing to their PFD. The Geriatric Pelvic Floor Rehab course can enlighten therapists on a score of comorbidities and techniques for handling those patients who are not sporting a marathon finisher medal to their physical therapy visits!
Erekson, E. A., Fried, T. R., Martin, D. K., Rutherford, T. J., Strohbehn, K., & Bynum, J. P. W. (2015). Frailty, cognitive impairment, and functional disability in older women with female pelvic floor dysfunction. International Urogynecology Journal, 26(6), 823–830. http://doi.org/10.1007/s00192-014-2596-2
K. Silay, S. Akinci, A. Ulas, A. Yalcin, Y.S. Silay, M.B. Akinci, I. Dilek, B. Yalcin. (2016). Occult urinary incontinence in elderly women and its association with geriatric condition. European Review for Medical and Pharmacological Sciences. 20(3): 447-451.
When my 6 year old daughter ran to the bathroom 3-4 times before she got on the school bus every morning, I wasn’t too concerned, but I definitely took note. The day she was in tears and wouldn’t get off the toilet because she felt like she was still wet, I got worried (although slightly intrigued). No matter how much she wiped, she still felt wet. When she stood up, she felt like she was going to pee herself, making my sweet-natured girl slip into hysterics. After eliminating small amounts of urine 8 separate times in 3 hours and saying it burned, I assumed she had a urinary tract infection (UTI). A simple urine test ruled out UTI or diabetes (thankfully!). So then, what was my daughter’s diagnosis? The pediatrician simply referred to it as “a phase;” however, I had researched the symptoms before the visit.
 In 2014 Arlen et al. described a condition called “phantom urinary incontinence.” This refers to the situation when children experience the sensation of being wet (a presumptive urinary incontinence) when they are objectively dry. They considered 20 children (18 females, 2 males) referred to their pediatric urology clinic over a 5 year span, all who were all diagnosed with phantom urinary incontinence (PUI). The authors evaluated the concomitant diagnoses found among the boys and girls in the study. Lower urinary tract symptoms were present in 95% of the subjects. Associated bladder symptoms were found as well, with urgency in 75% and frequency in 50% of the children. Vaginitis occurred in 72% of the girls. Parents reported obsessive-compulsive disorder or obsessive-compulsive disorder personality traits in 70% of the children. In order to treat these patients, dietary modifications, timed voiding, and a bowel regimen were implemented to manage symptoms. A follow up at 14.4 months revealed 90% of the children’s bowel-bladder dysfunction improved and PUI resolved. The authors concluded children compliant with a rigid bladder-bowel regimen experience relief of their “phantom” incontinence as well as lower urinary tract symptoms, and a majority of PUI patients have obsessive-compulsive traits.
In 2014 Arlen et al. described a condition called “phantom urinary incontinence.” This refers to the situation when children experience the sensation of being wet (a presumptive urinary incontinence) when they are objectively dry. They considered 20 children (18 females, 2 males) referred to their pediatric urology clinic over a 5 year span, all who were all diagnosed with phantom urinary incontinence (PUI). The authors evaluated the concomitant diagnoses found among the boys and girls in the study. Lower urinary tract symptoms were present in 95% of the subjects. Associated bladder symptoms were found as well, with urgency in 75% and frequency in 50% of the children. Vaginitis occurred in 72% of the girls. Parents reported obsessive-compulsive disorder or obsessive-compulsive disorder personality traits in 70% of the children. In order to treat these patients, dietary modifications, timed voiding, and a bowel regimen were implemented to manage symptoms. A follow up at 14.4 months revealed 90% of the children’s bowel-bladder dysfunction improved and PUI resolved. The authors concluded children compliant with a rigid bladder-bowel regimen experience relief of their “phantom” incontinence as well as lower urinary tract symptoms, and a majority of PUI patients have obsessive-compulsive traits.
Oliver et al., (2013) studied how psychosocial comorbidities and body mass index relate to children with lower urinary tract dysfunction. Data on 358 patients with lower urinary tract dysfunction between 6 to 17 years old was collected, and the subjects’ parents completed questionnaires screening for lower urinary tract symptoms, stressful life events, and psychological comorbidities. Obesity was present in 28.5% of the children, 22.9% had a recent stress in life, and 22.9% had a psychiatric disorder. Under and overweight children, children with a recent life stressor, psychiatric disorder, or both, as well as the younger-aged children all had lower urinary tract symptom scores significantly higher than healthy weight subjects, those without psychosocial comorbidities, and older subjects. The results encourage screening for psychosocial issues and obesity in pediatric patients with lower urinary tract dysfunction.
Having read the research, I knew a life stressor was likely contributing to my daughter’s symptoms. I had already advised her to sit on the toilet every 1-2 hours, don’t let her bladder get too full, wipe gently from front to back, stop bubble baths, and wear looser pants. To conclude our $76 session, the doctor prescribed almost verbatim what my daughter had heard from me at home. Although thankful it wasn’t something more serious, I am curious what the diagnosis code is for “a phase” and when it will end.
Arlen, AM, Dewhurst, LL, Kirsch, SS, Dingle, AD, Scherz, HC, Kirsch, AJ. (2014). Phantom urinary incontinence in children with bladder-bowel dysfunction. Urology. 84(3):685-8. DOI: http://dx.doi.org/10.1016/j.urology.2014.04.046 Oliver, J.L., Campigotto, M.J., Coplen, D.E. et al,. (2013). Psychosocial comorbidities and obesity are associated with lower urinary tract symptoms in children with voiding dysfunction. The Journal of Urology. 190:1511–1515. DOI: http://dx.doi.org/10.1016/j.juro.2013.02.025
Perhaps you have seen the Facebook post by Alan Naughton (March 5, 2015) where a horse with one zebra leg tells another horse, “I can’t say I’m entirely pleased with my hip replacement.” Although this post makes some people laugh, I imagine surgical candidates cringe at the thought of complications. Few people hop onto a surgeon’s schedule with great enthusiasm. While hip replacements are sometimes inevitable for quality of life, other hip pathologies can be successfully treated with more conservative measures.
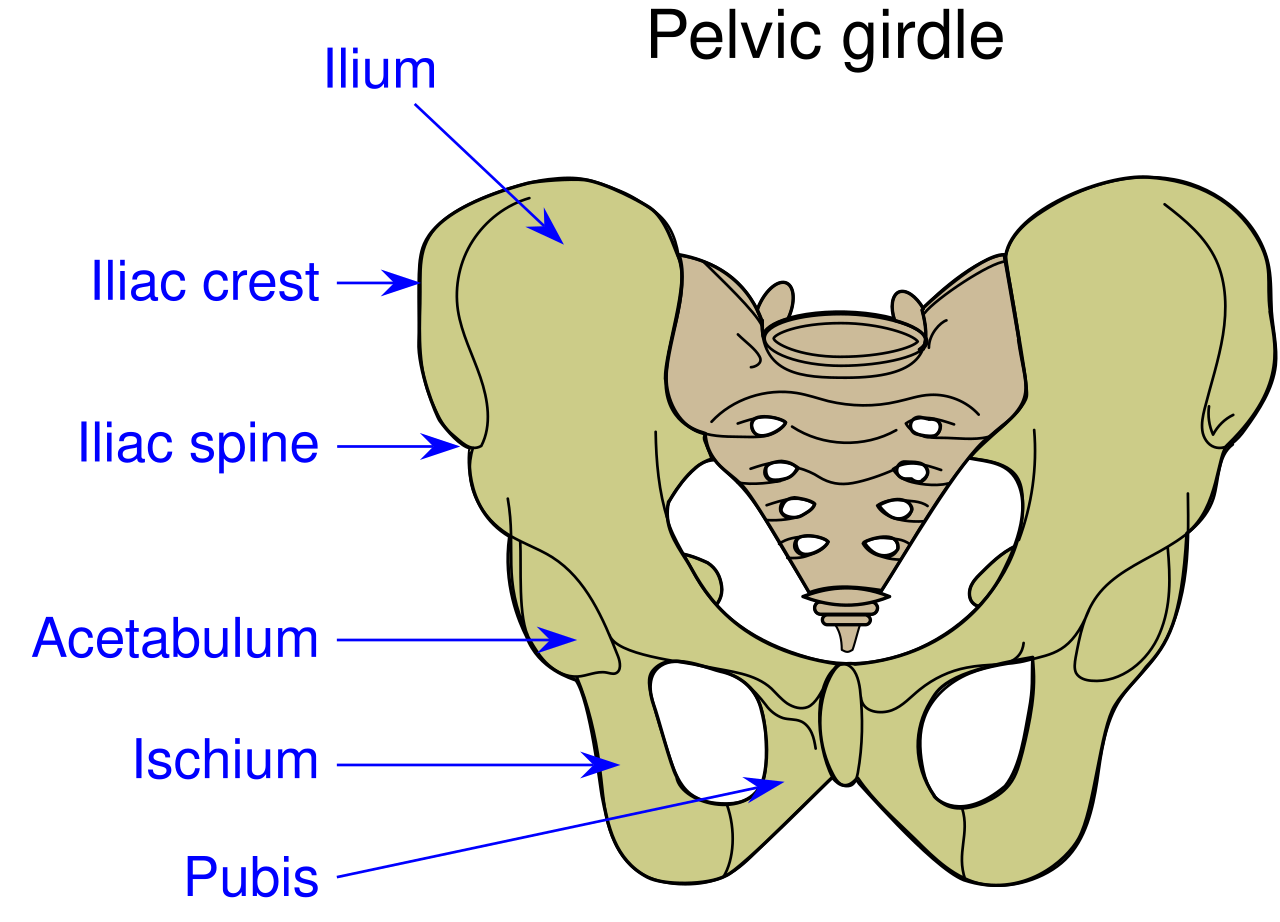 A case report in Manual Therapy (Lewis, Khuu, & Marinko 2015) described how postural correction and alternation of movement patterns were able to reduce hip pain secondary to acetabular dysplasia. A 31-year old female acute care nurse developed anterior hip pain with no trauma, and acetabular dysplasia as well as a labral tear were found. She got temporary relief of her constant ache and occasional sharp, intense pain from an intra-articular injection of cortisone. Her functional complaint was the pain prevented her from returning to recreational running. Intervention involved correcting the subject’s slight hip and knee hyperextension and posterior pelvic tilt with swayback posture, cueing her to walk on the treadmill with slight anterior pelvic tilt and contraction of the abdominals. This decreased her pain while walking from 6/10 to 2/10. Correction of the swayback posture decreased the hip flexion moment, decreasing stress on the anterior hip. At three months and then one year after the initial visit, she was relatively pain free. She still had pain with running, so she was advised to decrease her stride length and take shorter steps as well as decrease her hip extension by pushing off her feet more to minimize anterior hip joint reaction forces. With these cues, she was able to run without pain. Luckily for her, she had declined the option of acetabular reorientation surgery.
A case report in Manual Therapy (Lewis, Khuu, & Marinko 2015) described how postural correction and alternation of movement patterns were able to reduce hip pain secondary to acetabular dysplasia. A 31-year old female acute care nurse developed anterior hip pain with no trauma, and acetabular dysplasia as well as a labral tear were found. She got temporary relief of her constant ache and occasional sharp, intense pain from an intra-articular injection of cortisone. Her functional complaint was the pain prevented her from returning to recreational running. Intervention involved correcting the subject’s slight hip and knee hyperextension and posterior pelvic tilt with swayback posture, cueing her to walk on the treadmill with slight anterior pelvic tilt and contraction of the abdominals. This decreased her pain while walking from 6/10 to 2/10. Correction of the swayback posture decreased the hip flexion moment, decreasing stress on the anterior hip. At three months and then one year after the initial visit, she was relatively pain free. She still had pain with running, so she was advised to decrease her stride length and take shorter steps as well as decrease her hip extension by pushing off her feet more to minimize anterior hip joint reaction forces. With these cues, she was able to run without pain. Luckily for her, she had declined the option of acetabular reorientation surgery.
MacIntyre et al., (2015) presented a case study on conservative management of femoroacetabular impingement (FAI) in a retired 22 year old elite ice hockey goaltender. A 4-year history of left anterior hip pain forced him into early retirement. He was diagnosed with longitudinal acetabular labral tears with a cam-type FAI. Before considering surgery, he had to undergo physical therapy, which he did 1-2 times per week for 6 weeks. Treatment consisted of Active Release Technique (ART)® and soft tissue therapy with tools directed to the affected gluteal , iliopsoas, and adductor muscles and fascial planes, spinal manipulation of the right sacroiliac joint, left hip capsule distraction/release using the Mulligan concept, contemporary medical electroacupuncture, and extensive rehabilitation exercises for lumbopelvic stability. After 8 visits, he had no pain at rest or with exercise. At 8 weeks he returned to playing ice hockey and now plays competitively again with no need for surgery.
I would venture to guess no one who takes the conservative route for treatment of hip dysfunction comes out of physical therapy with irreconcilable side effects. Being able to skip surgery using manual therapy and postural correction is a huge goal. If you doubt you can treat the hip effectively, taking Manual Therapy for the Lumbo-Pelvic-Hip Complex can not only enhance your manual therapy approach to treatment but also introduce you to an exciting visual feedback system to maximize efficacy of core stabilization exercises.
Lewis, C. L., Khuu, A., & Marinko, L. (2015). Postural correction reduces hip pain in adult with acetabular dysplasia: a case report. Manual Therapy, 20(3), 508–512. http://doi.org/10.1016/j.math.2015.01.014 MacIntyre, K., Gomes, B., MacKenzie, S., & D’Angelo, K. (2015). Conservative management of an elite ice hockey goaltender with femoroacetabular impingement (FAI): a case report. The Journal of the Canadian Chiropractic Association, 59(4), 398–409.
Anxiety and depression are frequently encountered co-morbidities in the clients we serve in pelvic rehabilitation. This observation several years ago in clinical practice is one of many that prompted me down the path of exploring the connection between the gut, the brain, and overall health. In answering the question about these connections, I discovered many nutritionally related truths that are being rapidly elucidated in the literature.
A recent study by Sandhu, et.al. (2017) examines the role of the gut microbiota on the health of the brain and it’s influence on anxiety and depression. The title of the study, “Feeding the microbiota-gut-brain axis: diet, microbiome, and neuropsychiatry” gives us pause to consider the impact of our diets on this axis and in turn, on the health of our nervous system. The authors state:
It is diet composition and nutritional status that has been repeatedly been shown to be one of the most critical modifiable factors regulating the gut microbiota at different time points across the lifespan and under various health conditions.
With diet and nutritional status being the most critical modifiable factors in the health of this system, it becomes our responsibility to seek to understand this system and its influencing factors. We need to learn how to nourish the microbiota-gut-brain axis.
While anxiety and depression are common co-morbidities we encounter, we also commonly detect imbalance between the sympathetic and parasympathetic nervous system in our patients leading to, for example, pelvic floor muscle tension. In light of this study we must first and foremost ask: what is the microbiota? How can it influence our nervous system? How does this correlate to anxiety and depression? The answers to these questions provide clinical insight with far-reaching impact. We also consider: which circumstances disrupt the health of this system and which improve it? Finally, could understanding of this axis, among other nutritional correlates, provide a novel approach to bowel dysfunction, bladder dysfunction, chronic pelvic pain?
Be a part of the paradigm shift to integrative understanding as we explore these and many other burning questions. Please join us for insightful discussion in White Plains, NY March 31-April 1, 2017 for our next offering of Nutrition Perspectives for the Pelvic Rehab Therapist.
Sandhu, K. V., Sherwin, E., Schellekens, H., Stanton, C., Dinan, T. G., & Cryan, J. F. (2017). Feeding the microbiota-gut-brain axis: diet, microbiome, and neuropsychiatry. Transl Res, 179, 223-244. doi:10.1016/j.trsl.2016.10.002
In the comedy, Kindergarten Cop, Detective John Kimble may only have had a headache, not a tumor, but sometimes our patients do have a tumor. One of my patients was actually just diagnosed with a brain tumor after responding poorly to a cortisone injection for her neck pain. Tumors in other areas of the body, even in the pelvis, can be the source of symptoms that may seem like a nerve entrapment. This is a serious consideration to be given when diagnosing pudendal neuralgia.
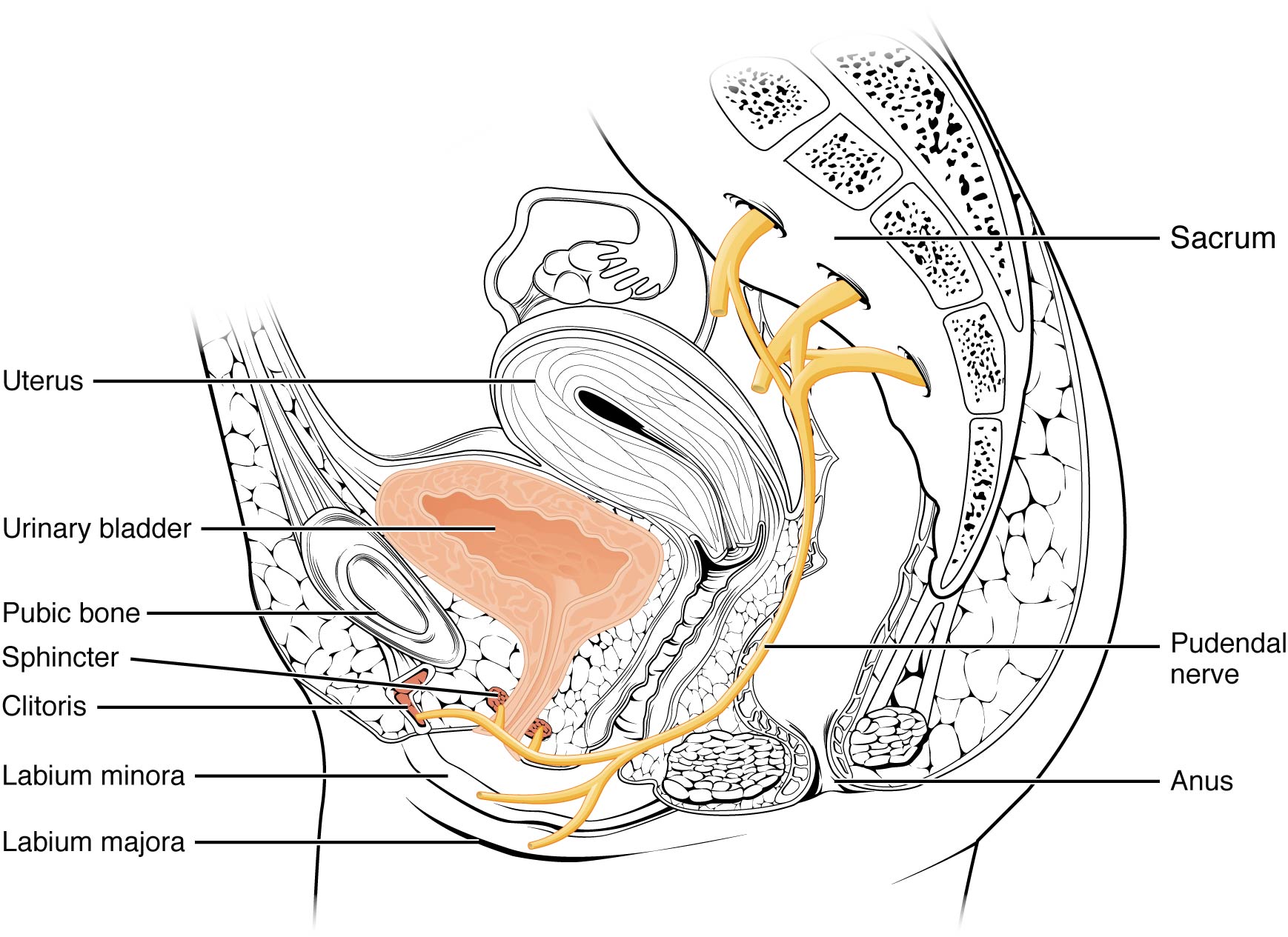 In 2008, Labat et al. published the “Diagnostic Criteria for Pudendal Neuralgia by Pudendal Nerve Entrapment” in Neurourology and Urodynamics . A group in Nantes, France, established criteria in 2006, since the diagnosis is primarily clinical in nature. The results of this paper concluded the five essential diagnostic criteria (Nantes criteria) are as follows:
In 2008, Labat et al. published the “Diagnostic Criteria for Pudendal Neuralgia by Pudendal Nerve Entrapment” in Neurourology and Urodynamics . A group in Nantes, France, established criteria in 2006, since the diagnosis is primarily clinical in nature. The results of this paper concluded the five essential diagnostic criteria (Nantes criteria) are as follows:
- Pain located in the anatomical region of the pudendal nerve.
- Pain worsened with sitting.
- Pain does NOT awaken the patient at night.
- Negative sensory loss upon clinical exam.
- Pain is relieved with an anesthetic pudendal nerve block.
A recent study by Waxweiler, Dobos, Thill, & Bruyninx explored the Nantes criteria as related to choosing surgical candidates for pudendal neuralgia from nerve entrapment. They looked at how a patient’s response to the anesthetic block corresponded to appropriate selection of patients for a successful surgical outcome. Six of 34 patients in the study had a negative anesthetic pudendal nerve block, and 100% of those patients had no symptom relief after surgery. In contrast, 64% of the patients who met all five of the Nantes criteria responded positively to surgery. The authors concluded confirmation of the 5th criteria as essential for predicting success of surgery for pudendal neuralgia by pudendal nerve entrapment.
In Pain Physician in 2016, Ploteau et al. present two case studies where consideration of the Nantes criteria helped diagnose rare tumors in patients who demonstrated red flags during examination. Warning signs such as nocturnal awakening, point-specific pain, pain of a neuropathic nature, and neurological deficits cannot be overlooked when a patient presents with pudendal neuralgia. In the case studies presented, the 31 year old woman did not have pain exacerbated with sitting and woke at night with pain, and the 62 year old woman was awakened at night with pain. Each patient had magnetic resonance imaging performed, and rare diagnoses of endometrial stromal sarcoma and adenoid cystic carcinoma were made, respectively. The tumors arose in the ischiorectal fossa and compressed the pudendal nerve, presenting as pudendal neuralgia in atypical forms requiring careful clinical examination and referral for MRI for accurate diagnosis.
Although a tumor rarely exists, it is our duty to recognize signs and symptoms that do not follow established criteria. Paying attention to what your patients say just may be lifesaving. Proper diagnosis of pudendal neuralgia is essential and sometimes falls in our hands.
Labat, JJ., Riant, T., Robert, R., Amarenco, G., Lefaucheur, JP., Rigaud, J. (2008). Diagnostic criteria for pudendal neuralgia by pudendal nerve entrapment (Nantes criteria). Neurourology and Urodynamics. 27(4):306-10. doi: 10.1002/nau.20505.
Waxweiler, C., Dobos, S., Thill, V., Bruyninx, L. (2016). Selection criteria for surgical treatment of pudendal neuralgia. Neurourology and Urodynamics. doi:10.1002/nau.22988.
Ploteau, S., Cardaillac, C., Perrouin-Verbe, M. , Riant, T., & Labat, J. (2016). Pudendal Neuralgia Due to Pudendal Nerve Entrapment: Warning Signs Observed in Two Cases and Review of the Literature. Pain Physician. 19:E449-E454.
How often have you heard that bedwetting was behavioral or caused by deep sleep and your child would outgrow it? 15% of children per year will “outgrow” bedwetting. What if your child is in the percentile at the end of that range?
Facts:

- Bedwetting affects 15% of girls and 22% of boys
- 5 - 7 Million US children
- Boys are 50% more likely than girls to wet the bed
- 10% of 6 year olds continue to wet
- Spontaneous cure rate 15% per year thereafter
- 1-3% of 18 year olds still wet their beds
- Less than 50% of all bedwetting children have bedwetting alone, without also experiencing daytime urinary leakage or constipation
- Bedwetting is genetic – if one parent was a bed wetter the child has a 40% chance of wetting the bed and if both parents were bedwetters the percentile goes up to 77%
Myths:
- Your child is lazy
- Your child is doing this to get attention
- Your child is just a deep sleeper
- You must wait to grow out of it
Research from the International Children’s Continence Society (ICCS) is a great resource for exploring the research on this topic and other pediatric voiding issues. www.i-c-c-s.org
What causes Bedwetting?
There are many philosophies discussed in the research. Here are some listed below:
- Hormone deficiency- our bladders empty about every 2-3 hours during the day however at night we can hold over 8 hours! This happens because our bodies produce an antidiuretic hormone when we sleep to slow kidney function and produce less urine to empty into the bladder. If this hormone is not being produced, the kidneys produce as much urine at night as they do during the day. In this case, it's good that the bladder empties out in our sleep, otherwise our bladders would be dangerously large and possibly reflux urine backward into the kidneys. Clearly not behavioral!!
- Dr. Steven Hodges has researched and written extensively on the topic of constipation causing pressure from the rectum against the bladder making it irritable during sleep. His research has supported the fact that once the bowel is cleaned out daily the bedwetting episodes diminish. See It’s No Accident by Dr. Hodges or visit https://www.bedwettingandaccidents.com for more information on this topic. Again, a physiological cause of bedwetting versus behavioral.
- Sleep Disturbance and Nasal Airway Obstruction. Dr. Neveus and colleagues reported that 43.5% of children with snoring or obstructive sleep apnea became dry after adenotonsillectomy. Dr. Kovacevic also found increases in antidiuretic hormone seen in responders post-operatively.
Take Home Message
- Active treatment for bedwetting should begin at age 6
- The impact of bedwetting is mainly psychological and may be severe
- Children with bedwetting have abnormal psychological test scores, however once the bedwetting is resolved the test scores return to normal
- “Treatment is not only justified but mandatory”
-ICCS Standardization document 2010
There is help!
At Physical Therapy Specialists we specialize in bedwetting, urinary leakage, constipation and other voiding issues in children. Let us eliminate the need for your family to suffer through this very treatable condition!
Al- Zaben FN, Sehlo MG. Punishement for bedwetting is associated with child depression and reduced quality of life. Child Abuse Negl. 2014
Hodges SJ, Colaco M. Daily enema regimen is superior to traditional therapies for nonneurogenic pediatric overactive bladder. Global Pediatric Health, 2016, 3: 1–4
Austin, P., Bauer, S.B., Bower, W., et al. The standardization of terminology of lower urinary tract function in children and adolescence: update report from the standardization committee of the international children’s continence society. J Urol (2014) 191.
Treatment response of an outpatient training for children with enuresis in a tertiary health care setting. J Pediatr Urol. 2012.
Hodges SJ,Anthony EY::aunrecognizedof. Urology.2012 Feb;79(2):421-4. doi: 10.1016/j.urology.2011.10.015. Epub 2011 Dec 14.
Kovacevic L, Wolfe-Christensen C, Lu H, Toton M, Mirkovic J, Thottam PJ, Abdulhamid I, Madgy D, Lakshmanan Y. Why does adenotonsillectomy not correct enuresis in all children with sleep disordered breathing? J Urol. 2014 May;191(5 Suppl):1592-6.
Nevéus T, Leissner L, Rudblad S, Bazargani F. Acta Paediatr. 2014 Jul 15. doi: 10.1111/apa.12749. [Epub ahead of print]Orthodontic widening of the palate may provide a cure for selected children with therapy-resistant enuresis.
Hodges, Steve J. It’s No Accident-Breakthrough solutions for your child’s wetting, constipation, UTI’s and other potty problems. © 2012. Lyons Press, Guilford, Connecticut.
My manual therapist husband once wrote a paper on the visceral referral pattern of the liver. Although he knows I injured my right shoulder shoveling snow a few years ago, whenever I have an exacerbation of shoulder pain, he likes to joke it is from my liver. (I would laugh if I had not acquired an affinity for red wine since having kids!) Sometimes pain in remote areas of our body really can be related to an organ in distress or simply “stuck” because of fascial restrictions around it. The kidneys in particular can refer pain into the low back and hips, and the bladder and ureters can provoke saddle area pain.
Tozzi, Bongiorno, and Vitturini (2012) looked into the kidney mobility of patients with low back pain. They used real-time Ultrasound to assess renal mobility before and after osteopathic fascial manipulation (OFM) via the Still Technique and Fascial Unwinding. The experimental group receiving OFM consisted of 109 people, and the control group receiving a sham treatment had 31 people, all with non-specific low back pain. For comparison, 101 subjects without back pain were also assessed with the ultrasound to determine a mean Kidney Mobility Score (KMS). The landmarks for measuring the renal mobility were the superior renal pole of the right kidney and the pillar of the right diaphragm, and they subtracted the distance at maximal inspiration (RdI) from that of maximal expiration (RdE). A significant difference was found in the KMS scores of asymptomatic versus symptomatic subjects with low back pain. Pre and post-RD values of the experimental group were significantly different from the control group. The short-form McGill Pain Questionnaire also demonstrated significant differences in the experimental versus control groups. The results of the study revealed a correlation between decreased renal mobility and non-specific low back pain and showed an improvement in renal mobility and low back pain after an osteopathic manipulation.
In 2016, Navot and Kalichman presented a case study of a 32 year old professional male cyclist with right hip and groin pain after an accident that caused a severe hip contusion and tearing of the tensor fascia latae and the gluteus medius muscles. A few rounds of physical therapy gave him partial relief of his pain in sitting and with cycling, and his hip range of motion only improved slightly. Despite no complaints of pelvic floor dysfunction, he was evaluated for involvement of the pelvic floor musculature and fascia. Pelvic Floor Fascial Mobilization was performed for 2 sessions, and the cyclist’s symptoms resolved completely. This case implied the efficacy of manual fascial release of the pelvic floor to reduce hip and groin pain.
When something seemingly orthopedic in nature does not respond with full resolution of symptoms from traditional physical therapy, the source of the pain may be deeper. Often times, we just need to ask the right questions to uncork the mystery of why a pain is lingering. No matter how skilled we are with our techniques, if we are not reaching the area in need, we are wasting our effort and our patients’ time and money. “Mobilization of Visceral Fascia: The Urinary System” is a course that provides a practitioner with the extra insight and tools to address potential sources of unresolved symptoms of low back, hip, and groin pain.
Tozzi, P., Bongiorno, D., and Vitturini, C. (2012) Low back pain and kidney mobility: local osteopathic fascial manipulation decreases pain perception and improves renal mobility. Journal of Bodywork and Movement Therapies. 16(3):381-91. doi: 10.1016/j.jbmt.2012.02.001
Navot, S and Kalichman, L. (2016). Hip and groin pain in a cyclist resolved after performing a pelvic floor fascial mobilization. Journal of Bodywork and Movement Therapies. 20(3):604-9. doi:10.1016/j.jbmt.2016.04.005
In this “quick fix” society, few people accept that musculoskeletal pain will require a commitment to following an exercise program for an extended period of time. If a hypomobile joint just needs to get moving and lubricated, one may get relief with a few manual therapy treatments and exercise sessions. However, if a joint is hypermobile (unstable) or degenerative and provokes a high level of pain, the rehab requires more time. The sacroiliac (SI) joint is one of those areas often requiring patients to work harder for the resolution of pain and dysfunction, but many seek surgical intervention instead.
 Polly et al. (2016) performed a randomized controlled trial of minimally invasive sacroiliac joint fusion (SIJF) with placement of a system of triangular titanium implants using a lateral transiliac approach versus non-surgical management (NSM) for SI dysfunction. Of the 148 subjects, 102 underwent SIJF and 46 had NSM. The NSM group received medication, physical therapy per American Physical Therapy Association guidelines, steroid injections and radiofrequency ablation of sacral nerve root lateral branches. The surgical group showed superior outcomes at a 2 year follow up, as clinical improvement per VAS pain score was 83.1% and ODI was 68.2%. The NSM group showed <10% improvement.
Polly et al. (2016) performed a randomized controlled trial of minimally invasive sacroiliac joint fusion (SIJF) with placement of a system of triangular titanium implants using a lateral transiliac approach versus non-surgical management (NSM) for SI dysfunction. Of the 148 subjects, 102 underwent SIJF and 46 had NSM. The NSM group received medication, physical therapy per American Physical Therapy Association guidelines, steroid injections and radiofrequency ablation of sacral nerve root lateral branches. The surgical group showed superior outcomes at a 2 year follow up, as clinical improvement per VAS pain score was 83.1% and ODI was 68.2%. The NSM group showed <10% improvement.
Sachs et al. (2016) studied outcomes of patients ≥3 years after SIJF for chronic (>5 years) SIJ dysfunction secondary to degenerative sacroiliitis or SIJ disruption. One hundred and seven patients participated in the study, and minimally invasive transiliac SIJF was definitively correlated with decreased pain, low disability scores, and improvements in activities of daily living performance. Sadly, these authors stated, “there is no high-quality evidence that physical therapy is effective in chronic SIJ pain.”
Even radiofrequency neurotomy or neural ablation revealed positive results for patients according to Reddy et al. (2016). The authors explored 14 patients’ responses 1 year after Simplicity radiofrequency (RF) of the lateral branches of S1-S3 in a retrospective review. Improvements in global health per SF12 as well as pain reduction were statistically significant.
Jonely et al. (2015) presented a case study of a woman with a 14-year history of SIJ pain whose symptoms persisted after 2 months of physical therapy. A multi-modal approach was then pursued with success, even at the 1 year follow up. The patient received 4 prolotherapy injections, SIJ manipulation into nutation, a pelvic girdle belt, and specific stabilization exercises. Over a 12-month period, the patient had 20 physical therapy sessions. Her Oswestry Disability score improved from 34% to 14% at 6 months and was 0% at 1 year. Numeric pain scale rating improved to 4/10 at 6 months and 0/10 at 1 year. The authors concluded a multimodal approach can be successful to manage SIJ dysfunction.
Clearly, if quality of life is so poor a person cannot function because of SIJ pain and therapy has failed, surgery may be the only choice. I exhaust all conservative measures before I cry “uncle” for a surgical fix. Despite a paucity of literature on manual therapy and sacroiliac treatment, I know there are clinicians successfully treating patients with SI dysfunction. Taking the Sacroiliac Joint Evaluation and Treatment can broaden your scope of understanding the SI joint and how to provide the most effective treatment, possibly preventing more invasive techniques for patients.
Polly, D. W., Swofford, J., Whang, P. G., Frank, C. J., Glaser, J. A., Limoni, R. P., … and the INSITE Study Group. (2016). Two-Year Outcomes from a Randomized Controlled Trial of Minimally Invasive Sacroiliac Joint Fusion vs. Non-Surgical Management for Sacroiliac Joint Dysfunction. International Journal of Spine Surgery, 10, 28. http://doi.org/10.14444/3028
Sachs, D., Kovalsky, D., Redmond, A., Limoni, R., Meyer, S. C., Harvey, C., & Kondrashov, D. (2016). Durable intermediate-to long-term outcomes after minimally invasive transiliac sacroiliac joint fusion using triangular titanium implants. Medical Devices (Auckland, N.Z.), 9, 213–222. http://doi.org/10.2147/MDER.S109276
Anjana Reddy, V. S., Sharma, C., Chang, K.-Y., & Mehta, V. (2016). “Simplicity” radiofrequency neurotomy of sacroiliac joint: a real life 1-year follow-up UK data. British Journal of Pain, 10(2), 90–99. http://doi.org/10.1177/2049463715627287
Jonely, H., Brismée, J.-M., Desai, M. J., & Reoli, R. (2015). Chronic sacroiliac joint and pelvic girdle dysfunction in a 35-year-old nulliparous woman successfully managed with multimodal and multidisciplinary approach. The Journal of Manual & Manipulative Therapy, 23(1), 20–26. http://doi.org/10.1179/2042618614Y.0000000086
After greeting a patient referred for temporomandibular joint dysfunction, the conversation began with an outpouring of emotion over a failed bladder sling surgery that left the woman with significant chronic pain, causing her to clench her jaw all the time. No matter what I was to find objectively with the examination, there was no doubt the treatment had to extend beyond joint mobilization, soft tissue work, and exercise. This woman clearly saw her cup as half empty, so filling her mind with a new approach to thinking about and dealing with her pain was essential for relieving her secondary jaw pain.
 Su et al. published a study called, “Pain Perception Can Be Modulated by Mindfulness Training: A Resting-State fMRI Study” (2016). The pain-afflicted group had 18 participants while the control group had 16. Brain behavior response of all subjects was measured per resting-state functional magnetic resonance imaging and 3 forms (Dallas Pain Questionnaire, Short Form McGill Pain Questionnaire-SFMPQ, and Kentucky Inventory of Mindfulness) before and after 6 weeks of mindfulness-based stress reduction treatment. Training consisted of mindfulness meditations such as a body scan, hatha yoga, walking and sitting meditation, and instruction on how to use the methods for pain management. After six 2.5-hour sessions/week and one 8-hour non-verbal session in the 4th week, the fMRI showed an increased connection from the anterior insular cortex (AIC) to the dorsal anterior midcingulate cortex (daMCC), and the SFMPQ scores were significantly improved in the pain-afflicted group. The authors suggested mindfulness training can change the brain connectivity responsible for our perception of pain.
Su et al. published a study called, “Pain Perception Can Be Modulated by Mindfulness Training: A Resting-State fMRI Study” (2016). The pain-afflicted group had 18 participants while the control group had 16. Brain behavior response of all subjects was measured per resting-state functional magnetic resonance imaging and 3 forms (Dallas Pain Questionnaire, Short Form McGill Pain Questionnaire-SFMPQ, and Kentucky Inventory of Mindfulness) before and after 6 weeks of mindfulness-based stress reduction treatment. Training consisted of mindfulness meditations such as a body scan, hatha yoga, walking and sitting meditation, and instruction on how to use the methods for pain management. After six 2.5-hour sessions/week and one 8-hour non-verbal session in the 4th week, the fMRI showed an increased connection from the anterior insular cortex (AIC) to the dorsal anterior midcingulate cortex (daMCC), and the SFMPQ scores were significantly improved in the pain-afflicted group. The authors suggested mindfulness training can change the brain connectivity responsible for our perception of pain.
Chadi et al.2016 performed a pilot study of female adolescents with chronic pain regarding the efficacy of mindfulness-based treatment. The experimental group (n=10) and the wait-list control group (n=9) consisted of girls between the ages of 13 and 18. For 8 weeks they met for a 90 minute session led by a psychiatry resident. Some of the mindfulness practices in this study included body scan, sitting and walking meditations, love and kindness meditations, mindful eating, compassion and deep listening, and breathing exercises. The wait-list control group also completed the 8-week program. Although all participants reported a positive change in the way they coped with pain, no statistically significant changes in quality of life, depression, anxiety, pain perception, and psychological distress were found. Significant salivary cortisol level improvements were observed (p<0.001) post mindful-based treatment session, indicating feasibility in pursuing further research with a larger randomized controlled trial.
Panahi and Faramarzi2016 studied mindfulness therapy effects on anxiety and depression for premenstrual syndrome (PMS). Sixty students (30 experimental, 30 control with no treatment) with mild to moderate PMS with depression underwent 8 weekly 120 minute sessions of mindfulness-based cognitive therapy (MBCT). Mean score improvements in depression, anxiety, and PMS were statistically significant from pre to post treatment for the subjects receiving MBCT. The authors stated MBCT psychotherapy could be considered beneficial for depression in mild to moderate PMS.
If jaw-clenching chronic pain owns a patient, he or she could benefit from managing the relationship through mindfulness. Our perception of pain is at the core of “whole body” treatment. The Mindfulness Based Pain Treatment course could help fill your patients’ as well as your own cup with healing.
If you're interested in learning more about mindfulness-based treatment techniques, Herman & Wallace offers three courses which you should consider. Mindfulness-Based Pain Treatment focuses on patient treatment, and the Mindfulness for Rehabilitation Professionals.
Su, I.-W., Wu, F.-W., Liang, K.-C., Cheng, K.-Y., Hsieh, S.-T., Sun, W.-Z., & Chou, T.-L. (2016). Pain Perception Can Be Modulated by Mindfulness Training: A Resting-State fMRI Study. Frontiers in Human Neuroscience, 10, 570. http://doi.org/10.3389/fnhum.2016.00570
Chadi, N., McMahon, A., Vadnais, M., Malboeuf-Hurtubise, C., Djemli, A., Dobkin, P. L., … Haley, N. (2016). Mindfulness-based Intervention for Female Adolescents with Chronic Pain: A Pilot Randomized Trial. Journal of the Canadian Academy of Child and Adolescent Psychiatry, 25(3), 159–168.
Panahi, F., & Faramarzi, M. (2016). The Effects of Mindfulness-Based Cognitive Therapy on Depression and Anxiety in Women with Premenstrual Syndrome. Depression Research and Treatment, 2016, 9816481. http://doi.org/10.1155/2016/9816481
The following comes to us from Carolyn McManus, PT, MS, MA, our resident expert in the power of mindfulness and it's applications to rehabilitation. Carolyn was recently featured in a video from the Journal of the American Medical Association for her contributions to a newly published research article. Join Carolyn at her course, Mindfulness Based Pain Treatment: A Biopsychosocial Approach to the Treatment of Chronic Pain on May 14th and 15th in California's Bay Area!
 Neuroimaging studies show that cortical and sub-cortical brain regions associated with cognitive and emotional processing connect directly with descending pain modulating circuits arising in the brainstem. As diminished nociceptive inhibition by descending pain modulation is a likely contributing factor to the persistence of pain, these cortical and sub-cortical connections to relevant brainstem regions provide a means by which maladaptive cognitive and emotional processing can contribute to the persistence of pain1. It is possible that strategies to help patients self-regulate cognitions and emotions could promote pain reduction through restoring the balance between excitatory and inhibitory mechanisms of the descending pain modulatory system.
Neuroimaging studies show that cortical and sub-cortical brain regions associated with cognitive and emotional processing connect directly with descending pain modulating circuits arising in the brainstem. As diminished nociceptive inhibition by descending pain modulation is a likely contributing factor to the persistence of pain, these cortical and sub-cortical connections to relevant brainstem regions provide a means by which maladaptive cognitive and emotional processing can contribute to the persistence of pain1. It is possible that strategies to help patients self-regulate cognitions and emotions could promote pain reduction through restoring the balance between excitatory and inhibitory mechanisms of the descending pain modulatory system.
To be mindful is to rest the mind in the present moment with stability and acceptance and without additional cognitive or emotional elaboration. Mindful body awareness is a central component. Training in mindful awareness has been shown to improve attention regulation, emotional processing and body awareness and contribute to reduced pain intensity, catastrophizing, depression and anxiety2,3,4,5. Training in mindfulness has also been shown to modulate brain activity in areas associated with body awareness and pain processing6,7. It is possible that the adaptive modulation of cortical and sub-cortical areas engaged with mindful cognitive, emotional and physical self-regulation could contribute to reducing pain through improving the balance between excitatory and inhibitory mechanisms of the descending pain modulatory system.
One of my patients reflected the clinical benefits of mindfulness training when he said, “I needed to learn how to not freak out when my exercises or daily activities increased my pain. Focusing my mind on the present moment was enormously helpful. I would tell myself, “Breathe. Just be here. Calm down.” By breathing and relaxing I could take control of how I was reacting and I immediately saw a difference. My pain did not increase out of control.”
I am thrilled to be sharing my 30+ year experience in mindfulness and patient care in my upcoming course through Herman and Wallace.
1. Ossipov M, Morimura K, Porreca F. Descending pain modulation and chronification of pain. Curr Opin Support Palliat Care 2014;8(2):143-151.
2. Holzel BK, Lazar SW, Guard T, et al. How does mindfulness meditation work? Proposing mechanisms of action from a conceptual and neural perspective. Perspect Psychol Science. 2011;6: 537–559.
3. Reiner K, Tibi L, Lipsitz JD. Do mindfulness-based interventions reduce pain intensity? A critical review of the literature. Pain Med. 2013 Feb;14(2):230-42.
4. Lakhan SE, Schofield KL. Mindfulness-based therapies in the treatment of somatization disorders: a systematic review and meta-analysis. PLoS One. 2013 Aug 26;8(8):e71834.
5. Schutze , Slater H, O’Sullivan P, et al. Mindfulness-based functional therapy: A preliminary open trial of an integrated model of care for people with persistent low back pain. Front Psychol. 2014 Aug 4;5:839.
6. Zeidan F, Martucci KT, Kraft RA, et al. Brain mechanisms supporting modulation of pain by mindfulness meditation. J Neurosci. 2011 Apr 6;31(14):5540-8.
7. Nakata H, Sakamoto K, Kakigi R. Meditation reduces pain-related activity in the anterior cingulated cortex, insula, secondary somatosensory cortex and thalamus. Front psychol. 2014;5:1489.















































